Zinc »
PDB 6o3r-6ofd »
6o7g »
Zinc in PDB 6o7g: Solution Structure of MLL4 PHD6 Domain in Complex with Histone H4K16AC Peptide
Enzymatic activity of Solution Structure of MLL4 PHD6 Domain in Complex with Histone H4K16AC Peptide
All present enzymatic activity of Solution Structure of MLL4 PHD6 Domain in Complex with Histone H4K16AC Peptide:
2.1.1.43;
2.1.1.43;
Zinc Binding Sites:
The binding sites of Zinc atom in the Solution Structure of MLL4 PHD6 Domain in Complex with Histone H4K16AC Peptide
(pdb code 6o7g). This binding sites where shown within
5.0 Angstroms radius around Zinc atom.
In total 2 binding sites of Zinc where determined in the Solution Structure of MLL4 PHD6 Domain in Complex with Histone H4K16AC Peptide, PDB code: 6o7g:
Jump to Zinc binding site number: 1; 2;
In total 2 binding sites of Zinc where determined in the Solution Structure of MLL4 PHD6 Domain in Complex with Histone H4K16AC Peptide, PDB code: 6o7g:
Jump to Zinc binding site number: 1; 2;
Zinc binding site 1 out of 2 in 6o7g
Go back to
Zinc binding site 1 out
of 2 in the Solution Structure of MLL4 PHD6 Domain in Complex with Histone H4K16AC Peptide
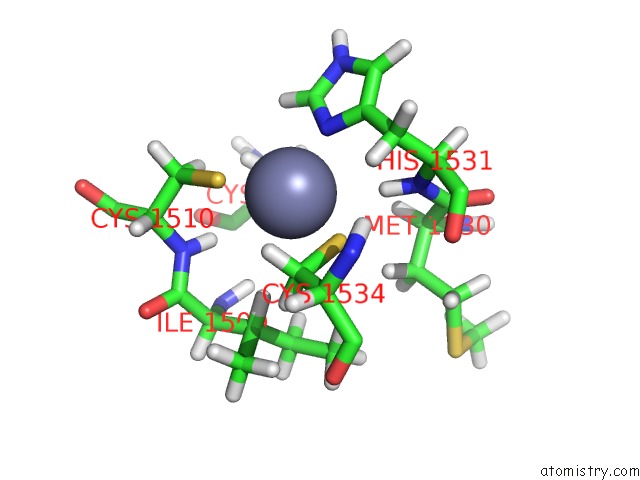
Mono view
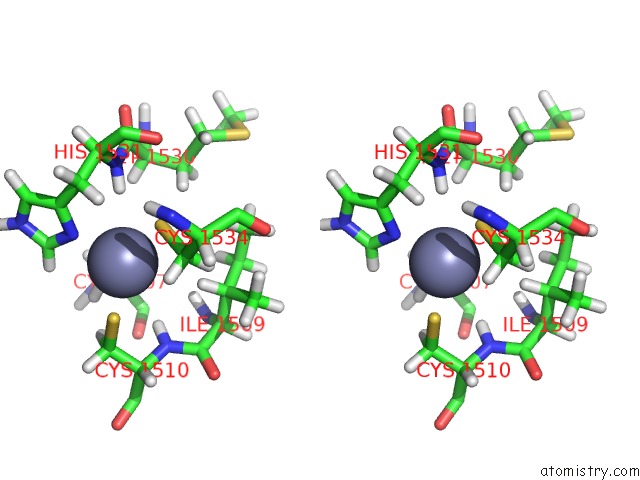
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 1 of Solution Structure of MLL4 PHD6 Domain in Complex with Histone H4K16AC Peptide within 5.0Å range:
|
Zinc binding site 2 out of 2 in 6o7g
Go back to
Zinc binding site 2 out
of 2 in the Solution Structure of MLL4 PHD6 Domain in Complex with Histone H4K16AC Peptide
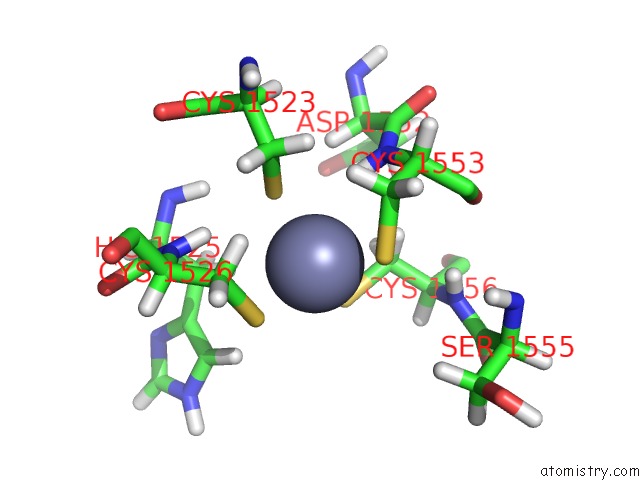
Mono view
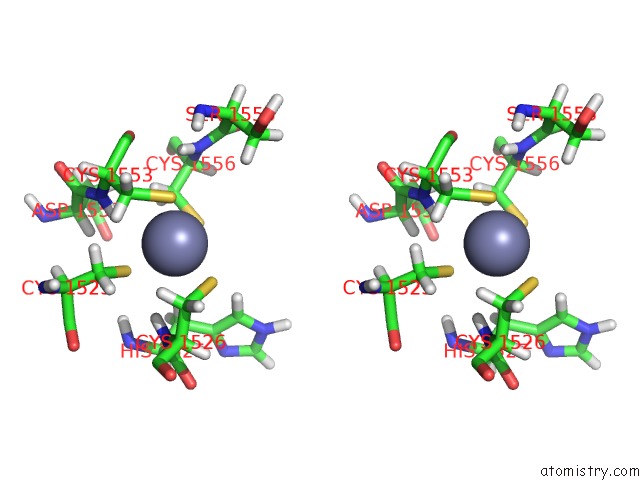
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 2 of Solution Structure of MLL4 PHD6 Domain in Complex with Histone H4K16AC Peptide within 5.0Å range:
|
Reference:
Y.Zhang,
Y.Jang,
J.E.Lee,
J.Ahn,
L.Xu,
M.R.Holden,
E.M.Cornett,
K.Krajewski,
B.J.Klein,
S.P.Wang,
Y.Dou,
R.G.Roeder,
B.D.Strahl,
S.B.Rothbart,
X.Shi,
K.Ge,
T.G.Kutateladze.
Selective Binding of the PHD6 Finger of MLL4 to Histone H4K16AC Links MLL4 and Mof. Nat Commun V. 10 2314 2019.
ISSN: ESSN 2041-1723
PubMed: 31127101
DOI: 10.1038/S41467-019-10324-8
Page generated: Tue Oct 29 04:07:06 2024
ISSN: ESSN 2041-1723
PubMed: 31127101
DOI: 10.1038/S41467-019-10324-8
Last articles
Zn in 9JYWZn in 9IR4
Zn in 9IR3
Zn in 9GMX
Zn in 9GMW
Zn in 9JEJ
Zn in 9ERF
Zn in 9ERE
Zn in 9EGV
Zn in 9EGW