Zinc »
PDB 9ere-9gkw »
9ere »
Zinc in PDB 9ere: SLFN11 Dimer Bound to Trna-Leu-Taa
Other elements in 9ere:
The structure of SLFN11 Dimer Bound to Trna-Leu-Taa also contains other interesting chemical elements:
| Magnesium | (Mg) | 4 atoms |
| Manganese | (Mn) | 3 atoms |
Zinc Binding Sites:
The binding sites of Zinc atom in the SLFN11 Dimer Bound to Trna-Leu-Taa
(pdb code 9ere). This binding sites where shown within
5.0 Angstroms radius around Zinc atom.
In total 2 binding sites of Zinc where determined in the SLFN11 Dimer Bound to Trna-Leu-Taa, PDB code: 9ere:
Jump to Zinc binding site number: 1; 2;
In total 2 binding sites of Zinc where determined in the SLFN11 Dimer Bound to Trna-Leu-Taa, PDB code: 9ere:
Jump to Zinc binding site number: 1; 2;
Zinc binding site 1 out of 2 in 9ere
Go back to
Zinc binding site 1 out
of 2 in the SLFN11 Dimer Bound to Trna-Leu-Taa
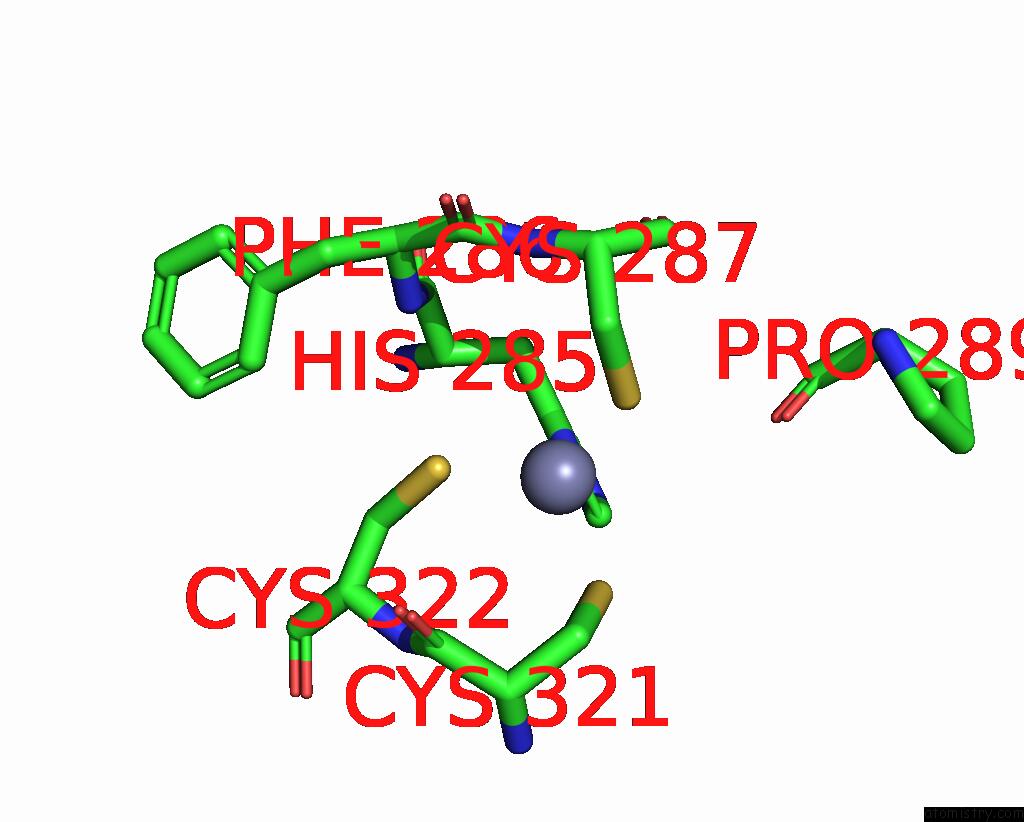
Mono view
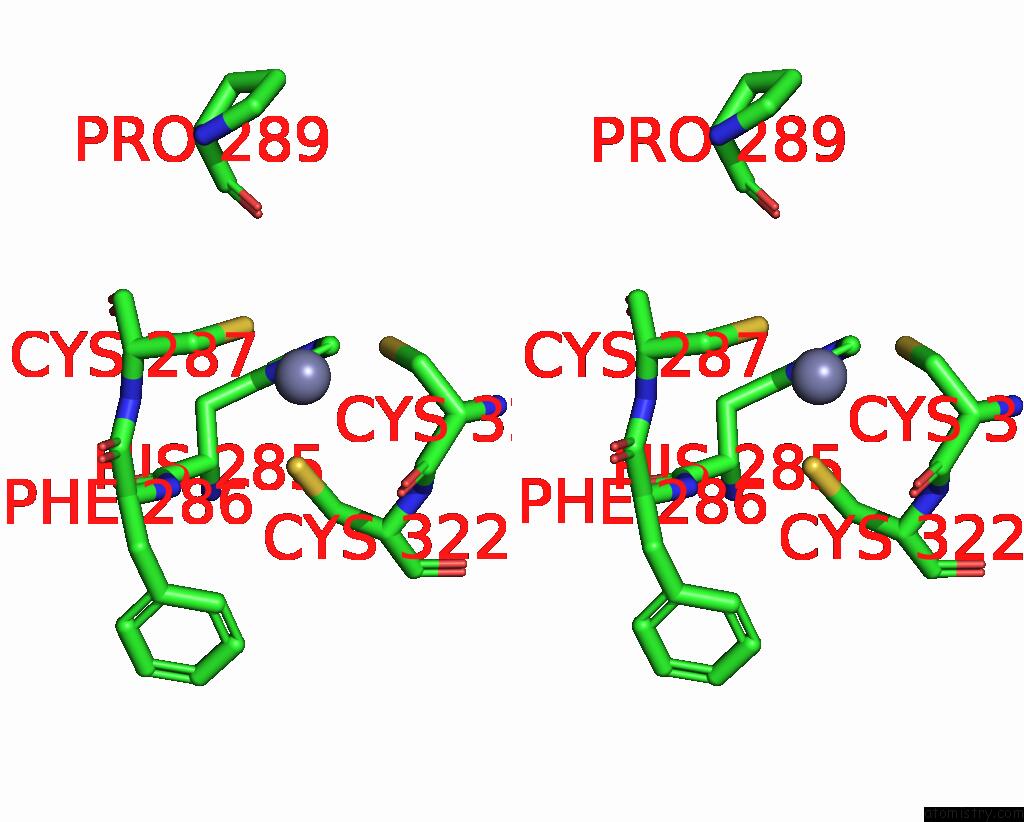
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 1 of SLFN11 Dimer Bound to Trna-Leu-Taa within 5.0Å range:
|
Zinc binding site 2 out of 2 in 9ere
Go back to
Zinc binding site 2 out
of 2 in the SLFN11 Dimer Bound to Trna-Leu-Taa
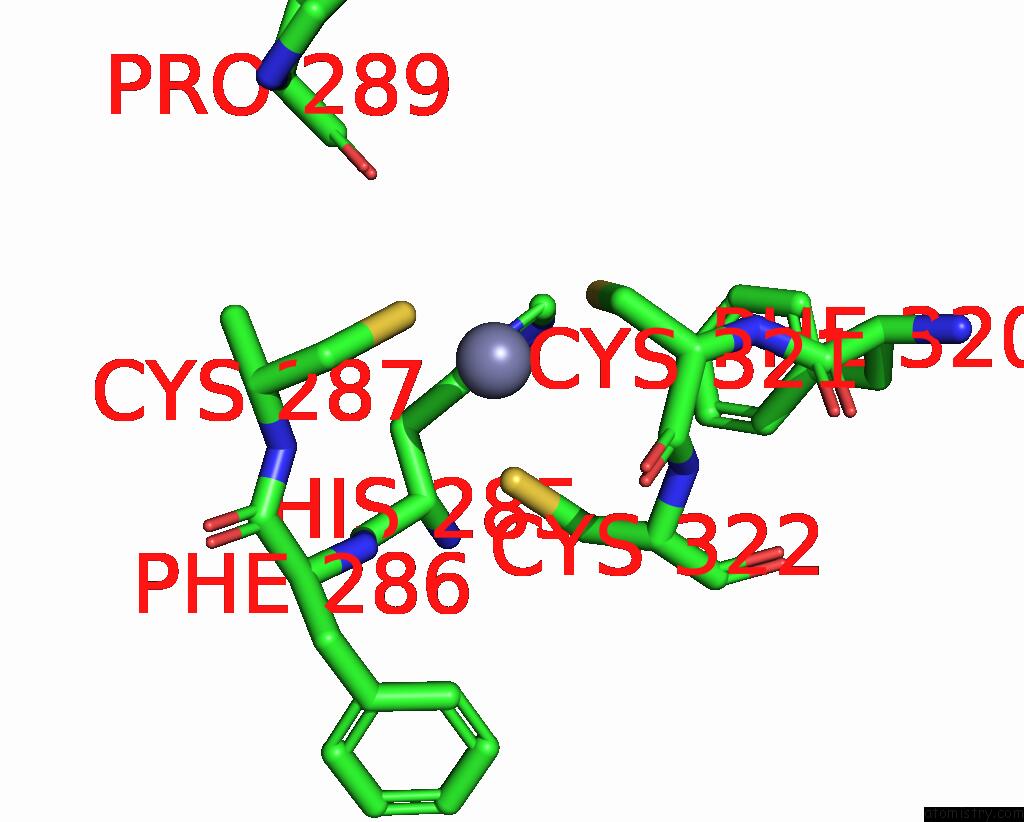
Mono view
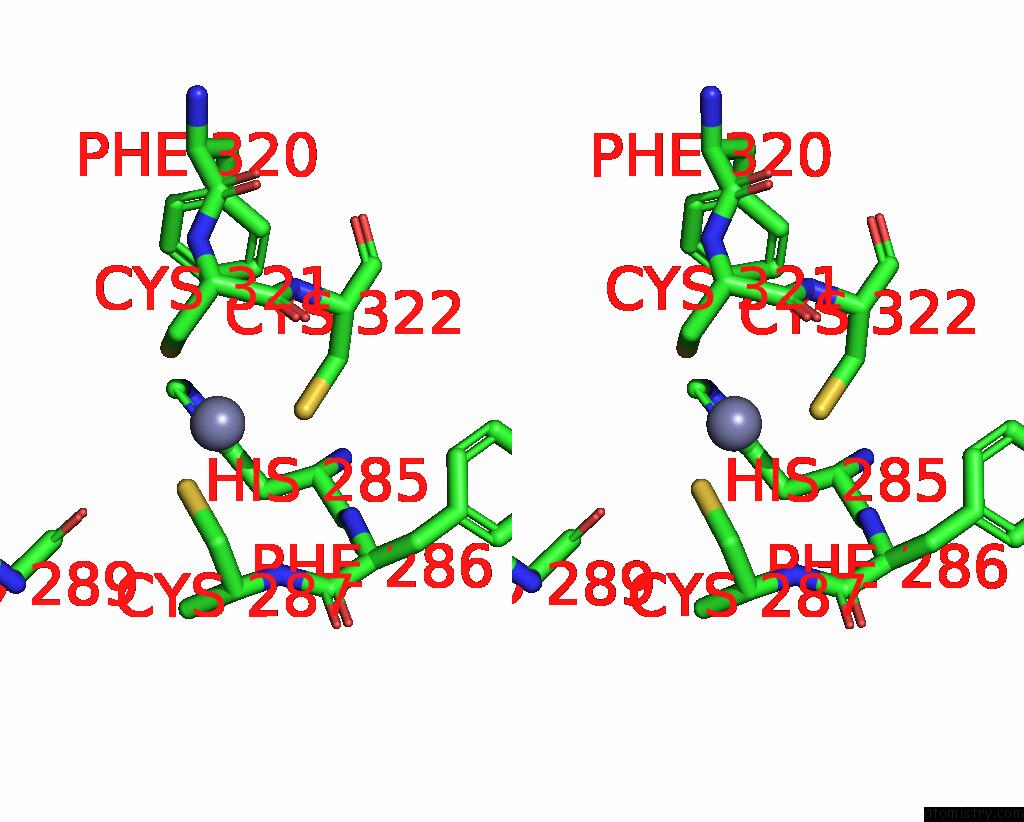
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 2 of SLFN11 Dimer Bound to Trna-Leu-Taa within 5.0Å range:
|
Reference:
M.Kugler,
F.J.Metzner,
K.P.Hopfner,
K.Lammens.
Phosphorylation-Mediated Conformational Change Regulates Human SLFN11 Nat Commun 2024.
ISSN: ESSN 2041-1723
DOI: 10.1038/S41467-024-54833-7
Page generated: Sun Dec 15 12:18:21 2024
ISSN: ESSN 2041-1723
DOI: 10.1038/S41467-024-54833-7
Last articles
Zn in 9JYWZn in 9IR4
Zn in 9IR3
Zn in 9GMX
Zn in 9GMW
Zn in 9JEJ
Zn in 9ERF
Zn in 9ERE
Zn in 9EGV
Zn in 9EGW