Zinc »
PDB 5msb-5n2x »
5n1j »
Zinc in PDB 5n1j: Crystal Structure of the Polysaccharide Deacetylase BC1974 From Bacillus Cereus
Protein crystallography data
The structure of Crystal Structure of the Polysaccharide Deacetylase BC1974 From Bacillus Cereus, PDB code: 5n1j
was solved by
P.Giastas,
A.Andreou,
S.Balomenou,
V.Bouriotis,
E.E.Eliopoulos,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 45.27 / 1.80 |
| Space group | P 1 21 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 50.280, 117.668, 99.768, 90.00, 102.75, 90.00 |
| R / Rfree (%) | 17.3 / 20.2 |
Zinc Binding Sites:
The binding sites of Zinc atom in the Crystal Structure of the Polysaccharide Deacetylase BC1974 From Bacillus Cereus
(pdb code 5n1j). This binding sites where shown within
5.0 Angstroms radius around Zinc atom.
In total 4 binding sites of Zinc where determined in the Crystal Structure of the Polysaccharide Deacetylase BC1974 From Bacillus Cereus, PDB code: 5n1j:
Jump to Zinc binding site number: 1; 2; 3; 4;
In total 4 binding sites of Zinc where determined in the Crystal Structure of the Polysaccharide Deacetylase BC1974 From Bacillus Cereus, PDB code: 5n1j:
Jump to Zinc binding site number: 1; 2; 3; 4;
Zinc binding site 1 out of 4 in 5n1j
Go back to
Zinc binding site 1 out
of 4 in the Crystal Structure of the Polysaccharide Deacetylase BC1974 From Bacillus Cereus
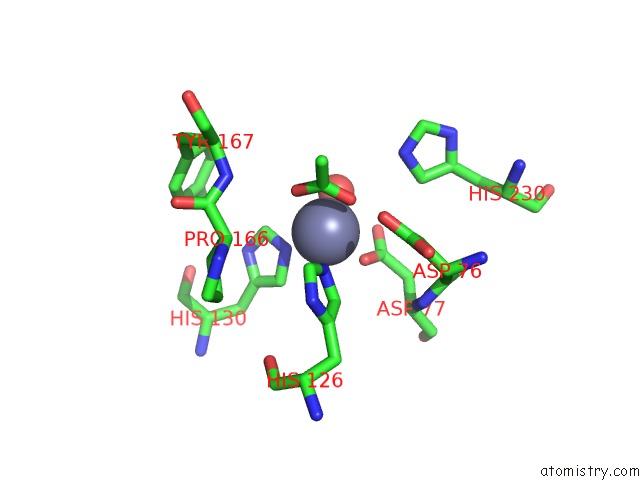
Mono view
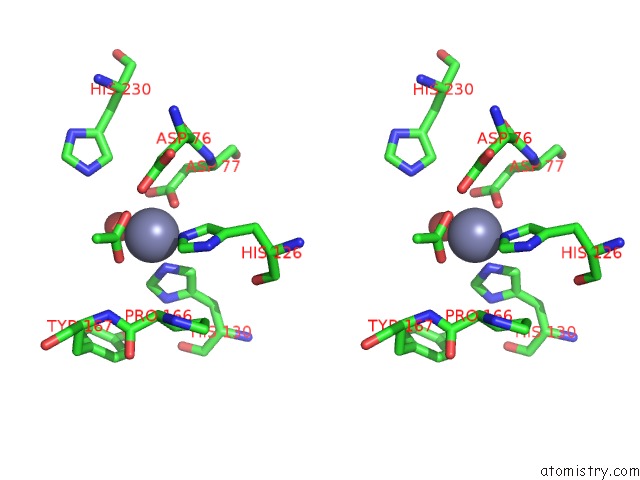
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 1 of Crystal Structure of the Polysaccharide Deacetylase BC1974 From Bacillus Cereus within 5.0Å range:
|
Zinc binding site 2 out of 4 in 5n1j
Go back to
Zinc binding site 2 out
of 4 in the Crystal Structure of the Polysaccharide Deacetylase BC1974 From Bacillus Cereus
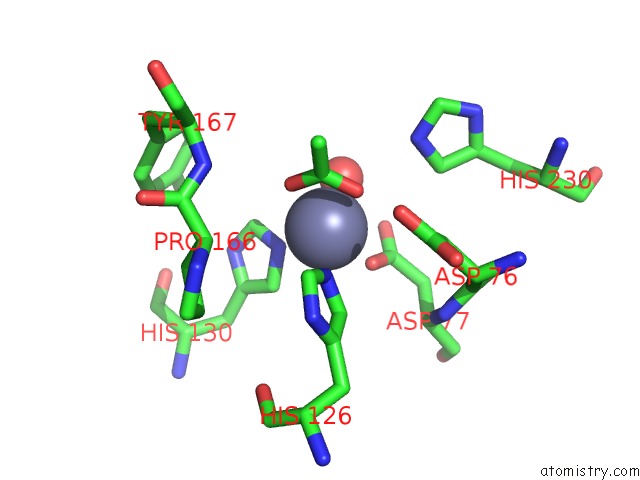
Mono view
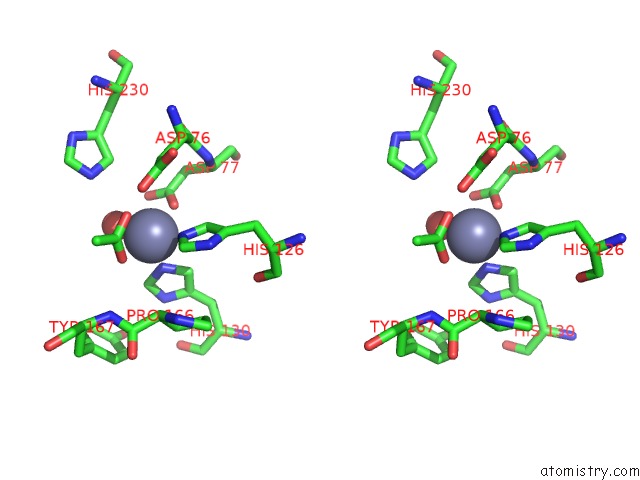
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 2 of Crystal Structure of the Polysaccharide Deacetylase BC1974 From Bacillus Cereus within 5.0Å range:
|
Zinc binding site 3 out of 4 in 5n1j
Go back to
Zinc binding site 3 out
of 4 in the Crystal Structure of the Polysaccharide Deacetylase BC1974 From Bacillus Cereus
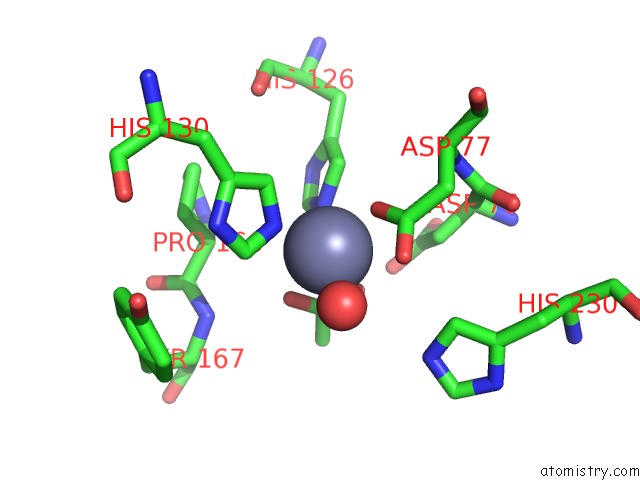
Mono view
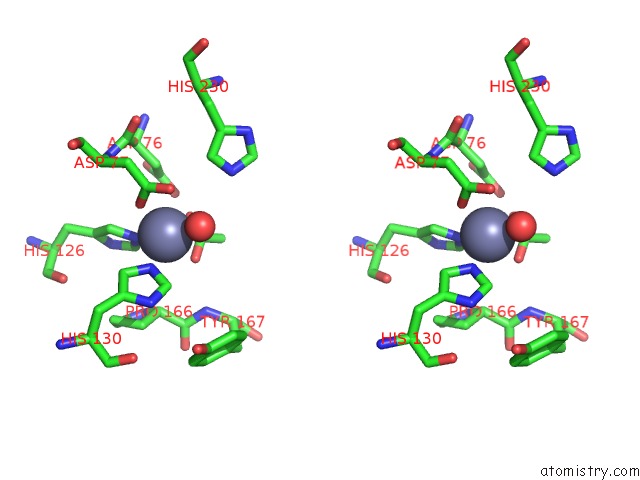
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 3 of Crystal Structure of the Polysaccharide Deacetylase BC1974 From Bacillus Cereus within 5.0Å range:
|
Zinc binding site 4 out of 4 in 5n1j
Go back to
Zinc binding site 4 out
of 4 in the Crystal Structure of the Polysaccharide Deacetylase BC1974 From Bacillus Cereus
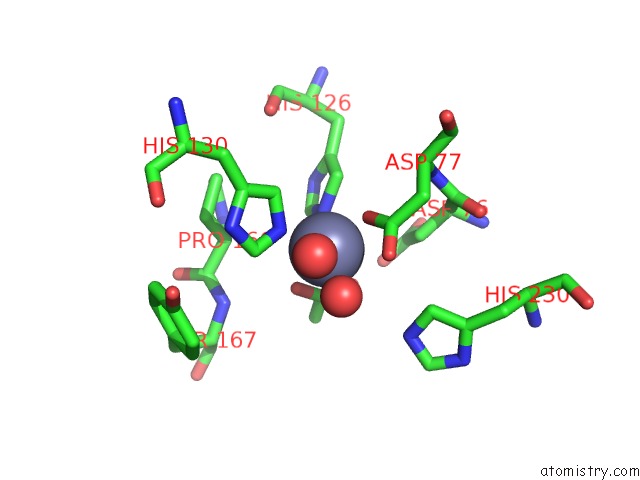
Mono view
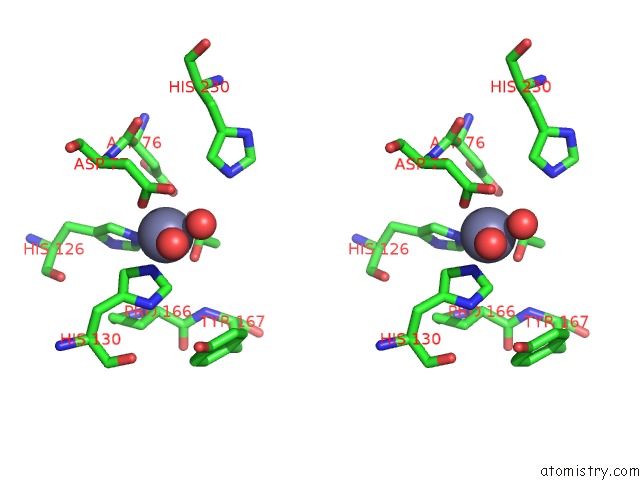
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 4 of Crystal Structure of the Polysaccharide Deacetylase BC1974 From Bacillus Cereus within 5.0Å range:
|
Reference:
P.Giastas,
A.Andreou,
A.Papakyriakou,
D.Koutsioulis,
S.Balomenou,
S.J.Tzartos,
V.Bouriotis,
E.E.Eliopoulos.
Structures of the Peptidoglycan N-Acetylglucosamine Deacetylase BC1974 and Its Complexes with Zinc Metalloenzyme Inhibitors. Biochemistry V. 57 753 2018.
ISSN: ISSN 1520-4995
PubMed: 29257674
DOI: 10.1021/ACS.BIOCHEM.7B00919
Page generated: Sun Oct 27 22:27:05 2024
ISSN: ISSN 1520-4995
PubMed: 29257674
DOI: 10.1021/ACS.BIOCHEM.7B00919
Last articles
Zn in 9JYWZn in 9IR4
Zn in 9IR3
Zn in 9GMX
Zn in 9GMW
Zn in 9JEJ
Zn in 9ERF
Zn in 9ERE
Zn in 9EGV
Zn in 9EGW