Zinc »
PDB 6iv1-6j4g »
6iv4 »
Zinc in PDB 6iv4: Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F
Protein crystallography data
The structure of Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F, PDB code: 6iv4
was solved by
S.Chen,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 61.93 / 3.14 |
| Space group | P 21 21 21 |
| Cell size a, b, c (Å), α, β, γ (°) | 104.009, 154.186, 161.736, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 23.8 / 29.5 |
Zinc Binding Sites:
Pages:
>>> Page 1 <<< Page 2, Binding sites: 11 - 13;Binding sites:
The binding sites of Zinc atom in the Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F (pdb code 6iv4). This binding sites where shown within 5.0 Angstroms radius around Zinc atom.In total 13 binding sites of Zinc where determined in the Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F, PDB code: 6iv4:
Jump to Zinc binding site number: 1; 2; 3; 4; 5; 6; 7; 8; 9; 10;
Zinc binding site 1 out of 13 in 6iv4
Go back to
Zinc binding site 1 out
of 13 in the Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F
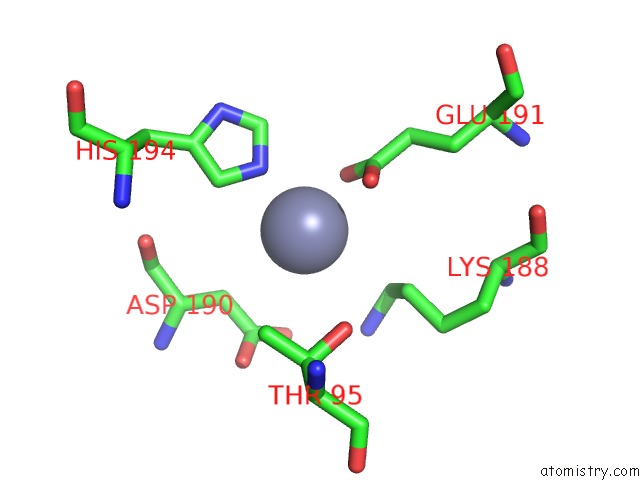
Mono view
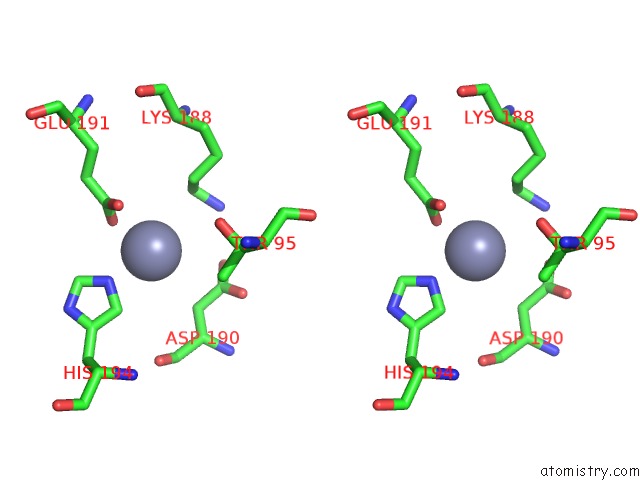
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 1 of Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F within 5.0Å range:
|
Zinc binding site 2 out of 13 in 6iv4
Go back to
Zinc binding site 2 out
of 13 in the Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F
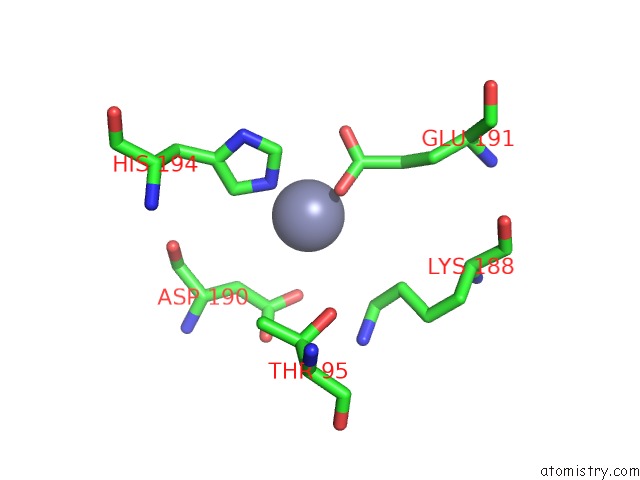
Mono view
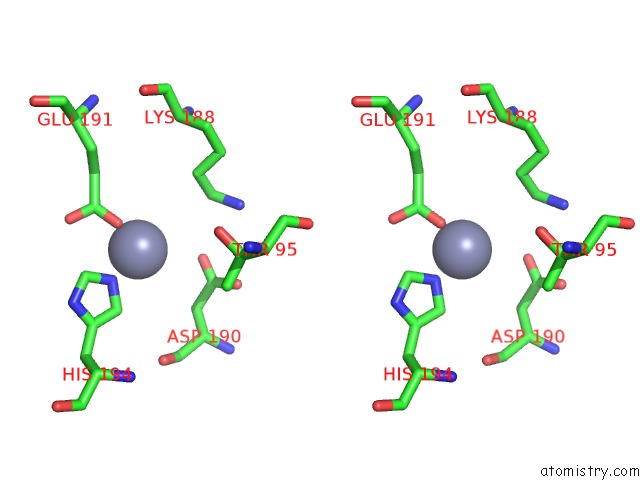
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 2 of Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F within 5.0Å range:
|
Zinc binding site 3 out of 13 in 6iv4
Go back to
Zinc binding site 3 out
of 13 in the Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F
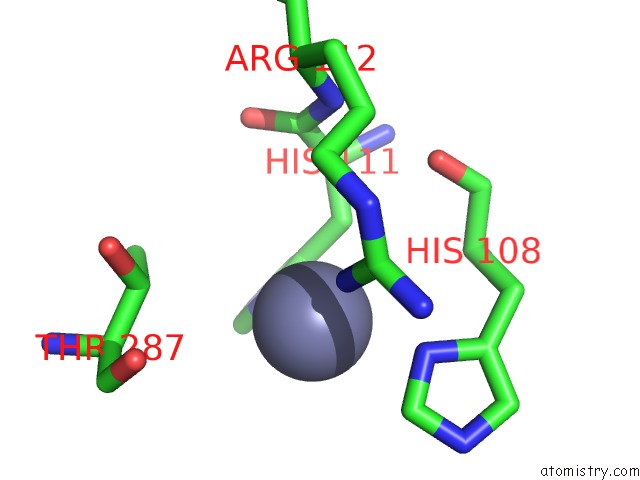
Mono view
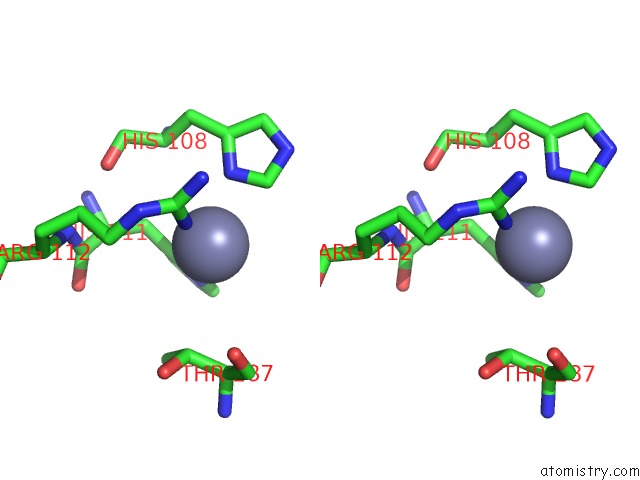
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 3 of Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F within 5.0Å range:
|
Zinc binding site 4 out of 13 in 6iv4
Go back to
Zinc binding site 4 out
of 13 in the Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F
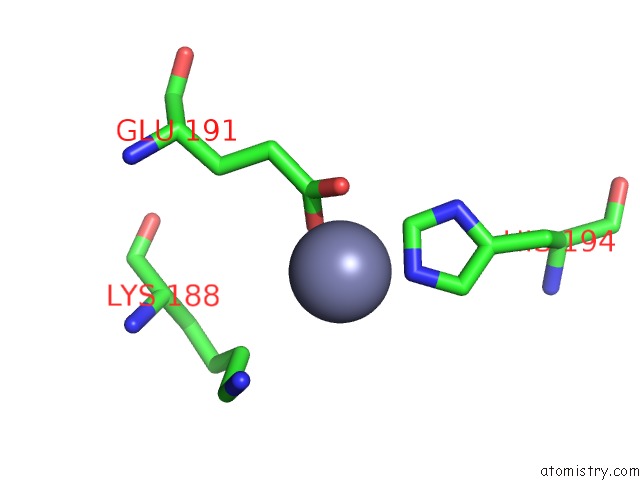
Mono view
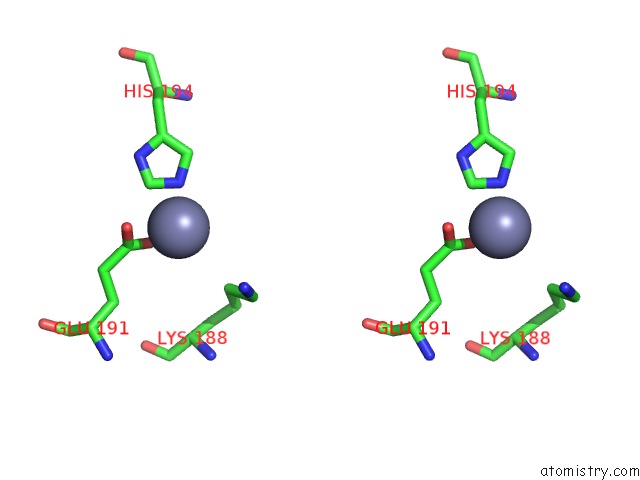
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 4 of Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F within 5.0Å range:
|
Zinc binding site 5 out of 13 in 6iv4
Go back to
Zinc binding site 5 out
of 13 in the Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F
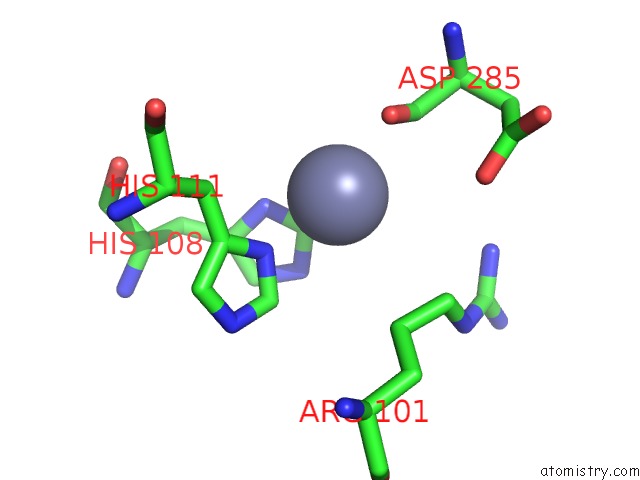
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 5 of Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F within 5.0Å range:
|
Zinc binding site 6 out of 13 in 6iv4
Go back to
Zinc binding site 6 out
of 13 in the Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F
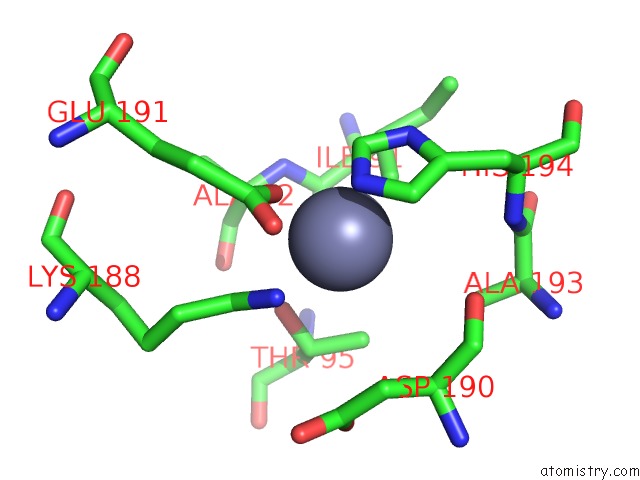
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 6 of Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F within 5.0Å range:
|
Zinc binding site 7 out of 13 in 6iv4
Go back to
Zinc binding site 7 out
of 13 in the Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F
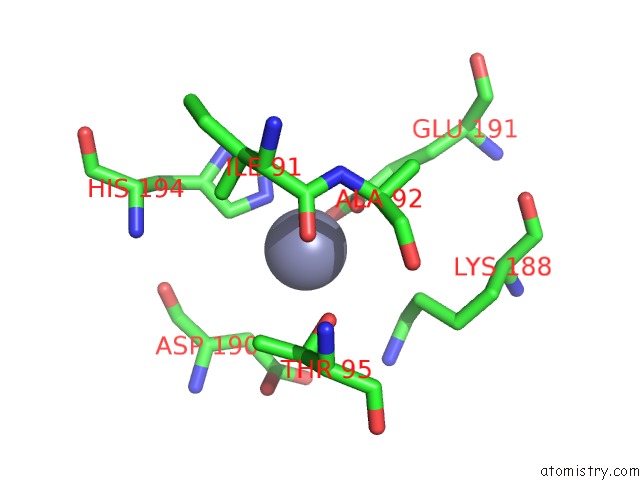
Mono view
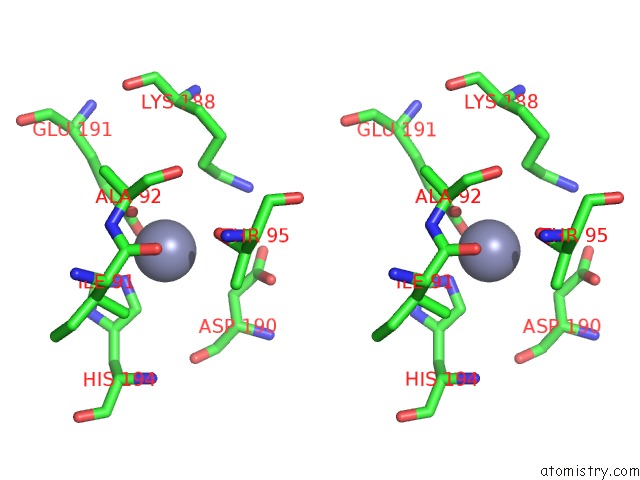
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 7 of Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F within 5.0Å range:
|
Zinc binding site 8 out of 13 in 6iv4
Go back to
Zinc binding site 8 out
of 13 in the Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F
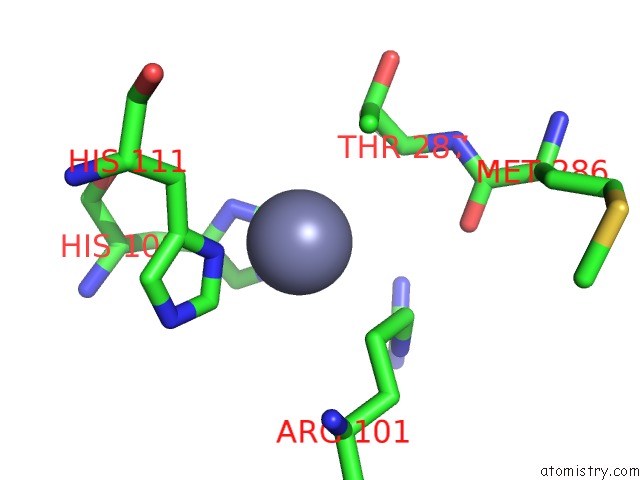
Mono view
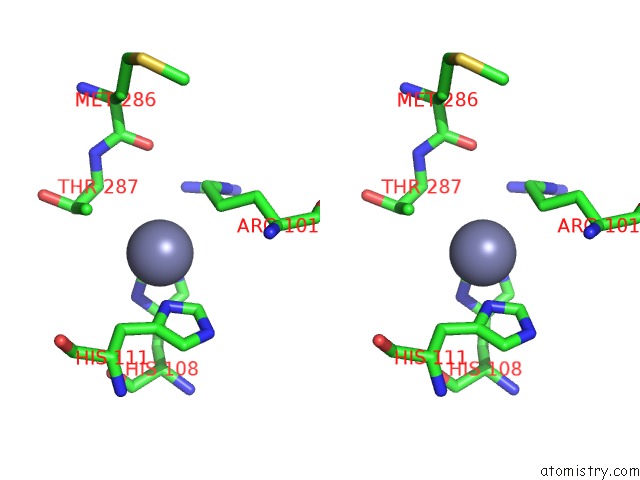
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 8 of Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F within 5.0Å range:
|
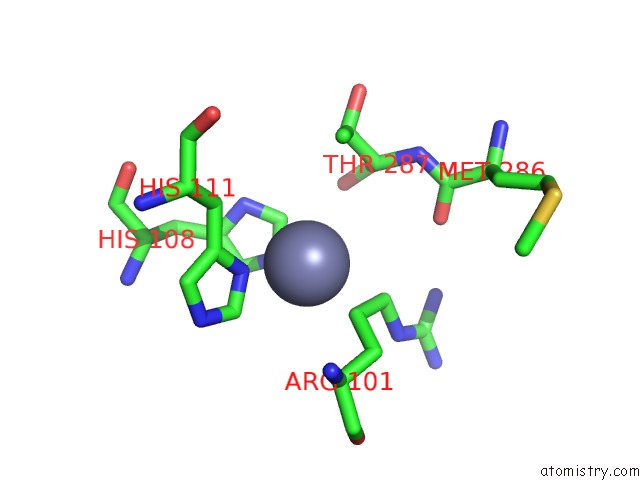
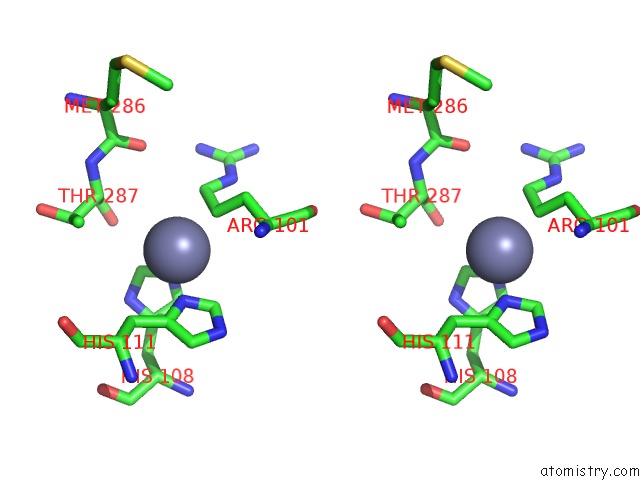
Zinc binding site 9 out of 13 in 6iv4
Go back to
Zinc binding site 9 out
of 13 in the Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 9 of Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F within 5.0Å range:
|
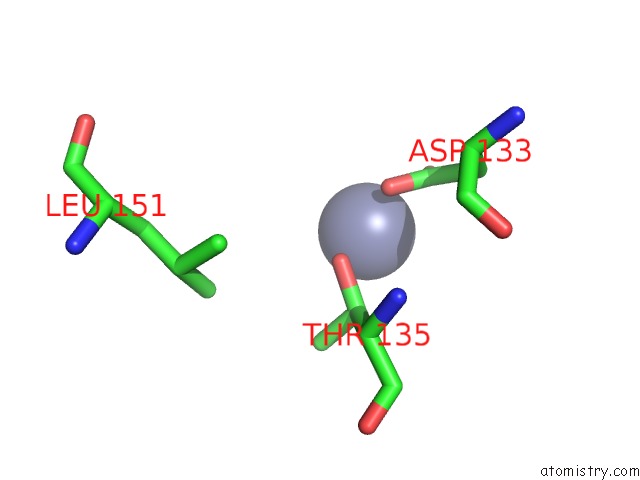
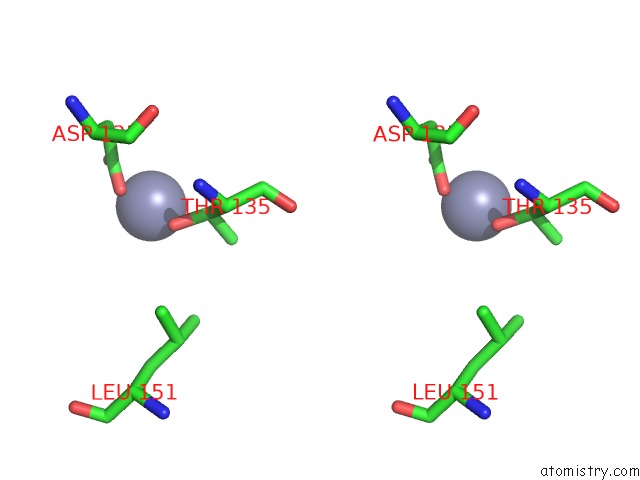
Zinc binding site 10 out of 13 in 6iv4
Go back to
Zinc binding site 10 out
of 13 in the Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 10 of Crystal Structure of A Bacterial Bestrophin Homolog From Klebsiella Pneumoniae with A Mutation W252F within 5.0Å range:
|
Reference:
C.Ji,
A.Kittredge,
A.Hopiavuori,
N.Ward,
S.Chen,
Y.Fukuda,
Y.Zhang,
T.Yang.
Dual CA2+-Dependent Gates in Human BESTROPHIN1 Underlie Disease-Causing Mechanisms of Gain-of-Function Mutations. Commun Biol V. 2 240 2019.
ISSN: ESSN 2399-3642
PubMed: 31263784
DOI: 10.1038/S42003-019-0433-3
Page generated: Thu Aug 21 15:57:26 2025
ISSN: ESSN 2399-3642
PubMed: 31263784
DOI: 10.1038/S42003-019-0433-3
Last articles
Zn in 6RNMZn in 6RMY
Zn in 6RMX
Zn in 6RMR
Zn in 6RMN
Zn in 6RMP
Zn in 6RMG
Zn in 6RIM
Zn in 6RLY
Zn in 6RM1