Zinc »
PDB 2b8k-2bnm »
2bg2 »
Zinc in PDB 2bg2: Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced.
Enzymatic activity of Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced.
All present enzymatic activity of Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced.:
3.5.2.6;
3.5.2.6;
Protein crystallography data
The structure of Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced., PDB code: 2bg2
was solved by
A.M.Davies,
R.M.Rasia,
A.J.Vila,
B.J.Sutton,
S.M.Fabiane,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 6.00 / 2.40 |
| Space group | P 31 2 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 67.300, 67.300, 177.183, 90.00, 90.00, 120.00 |
| R / Rfree (%) | 17.9 / 22.4 |
Other elements in 2bg2:
The structure of Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced. also contains other interesting chemical elements:
| Chlorine | (Cl) | 1 atom |
Zinc Binding Sites:
The binding sites of Zinc atom in the Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced.
(pdb code 2bg2). This binding sites where shown within
5.0 Angstroms radius around Zinc atom.
In total 4 binding sites of Zinc where determined in the Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced., PDB code: 2bg2:
Jump to Zinc binding site number: 1; 2; 3; 4;
In total 4 binding sites of Zinc where determined in the Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced., PDB code: 2bg2:
Jump to Zinc binding site number: 1; 2; 3; 4;
Zinc binding site 1 out of 4 in 2bg2
Go back to
Zinc binding site 1 out
of 4 in the Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced.
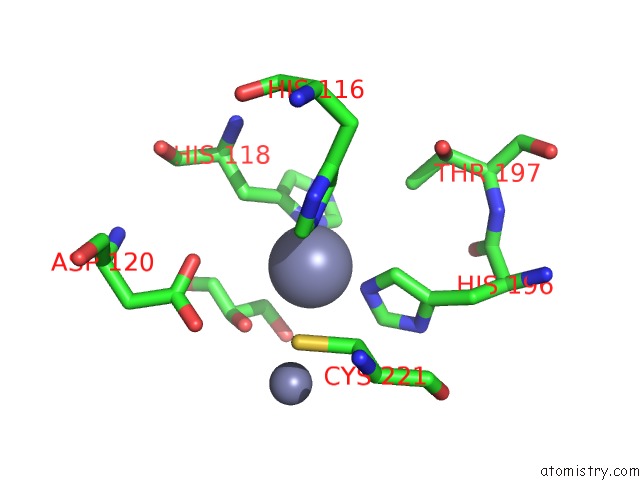
Mono view
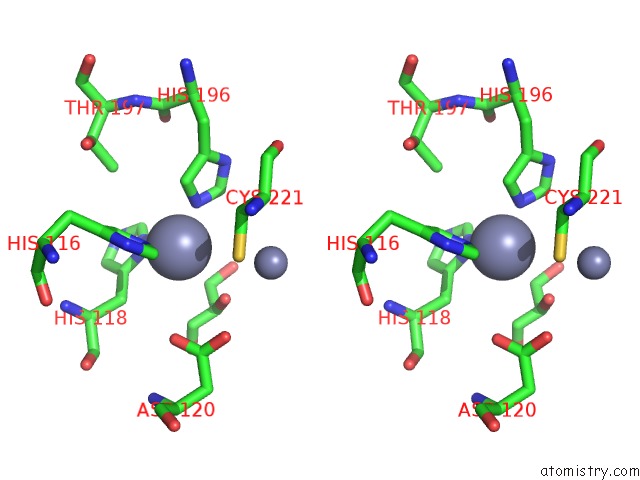
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 1 of Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced. within 5.0Å range:
|
Zinc binding site 2 out of 4 in 2bg2
Go back to
Zinc binding site 2 out
of 4 in the Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced.
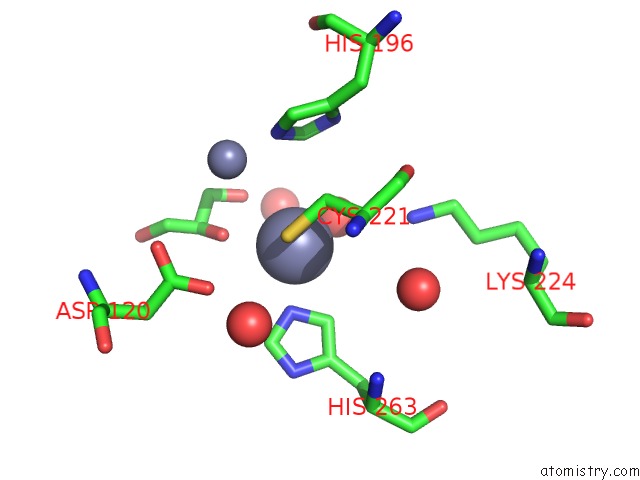
Mono view
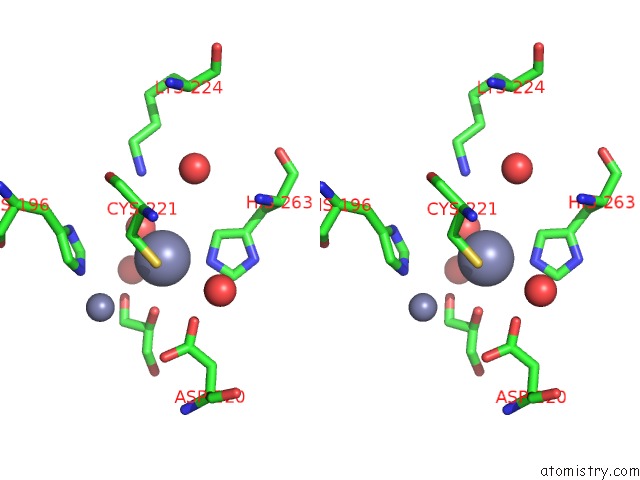
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 2 of Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced. within 5.0Å range:
|
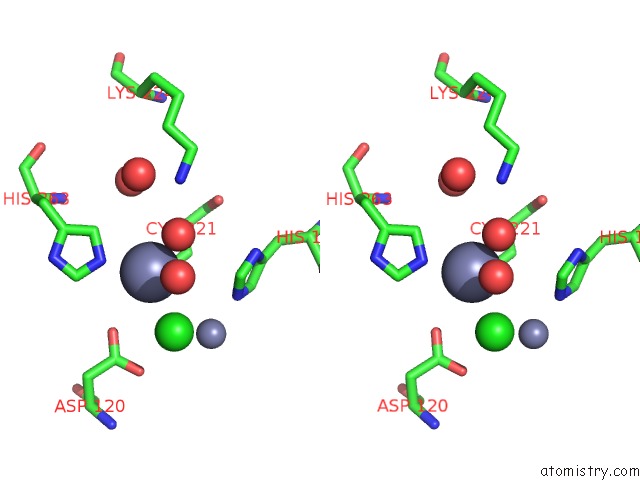
Zinc binding site 3 out of 4 in 2bg2
Go back to
Zinc binding site 3 out
of 4 in the Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced.
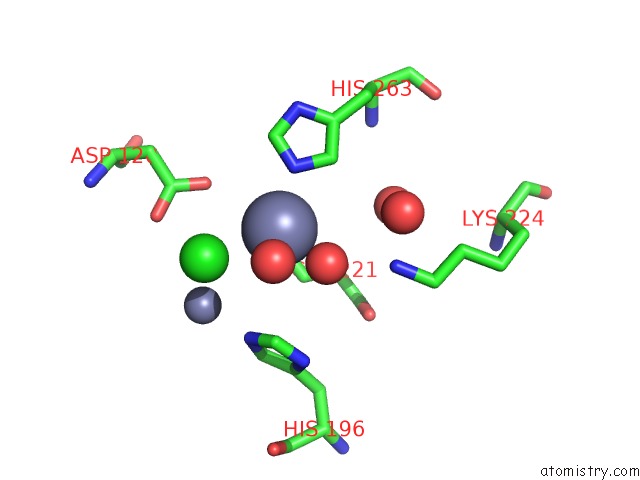
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 3 of Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced. within 5.0Å range:
|
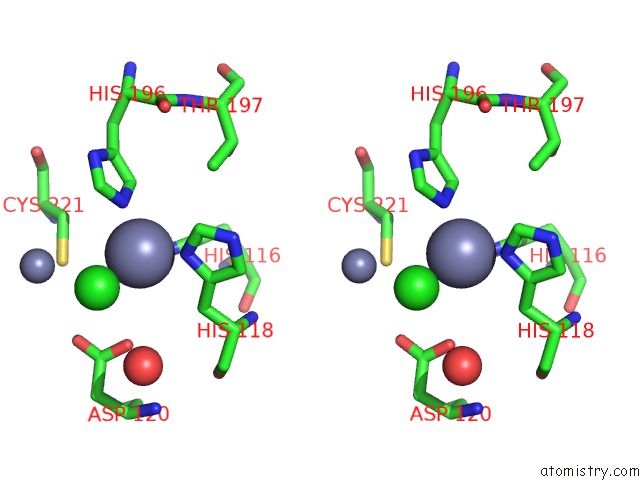
Zinc binding site 4 out of 4 in 2bg2
Go back to
Zinc binding site 4 out
of 4 in the Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced.
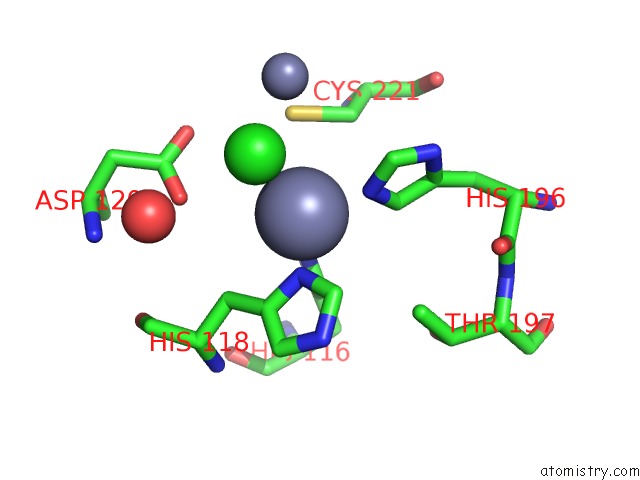
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 4 of Bacillus Cereus Metallo-Beta-Lactamase (Bcii) Arg (121) Cys Mutant. Solved at PH4.5 Using 20MM ZNSO4 in the Buffer. 1MM Dtt and 1MM Tcep- Hcl Were Used As Reducing Agents. CYS221 Is Reduced. within 5.0Å range:
|
Reference:
A.M.Davies,
R.M.Rasia,
A.J.Vila,
B.J.Sutton,
S.M.Fabiane.
Effect of pH on the Active Site of An ARG121CYS Mutant of the Metallo-Beta-Lactamase From Bacillus Cereus: Implications For the Enzyme Mechanism. Biochemistry V. 44 4841 2005.
ISSN: ISSN 0006-2960
PubMed: 15779910
DOI: 10.1021/BI047709T
Page generated: Wed Oct 16 22:00:36 2024
ISSN: ISSN 0006-2960
PubMed: 15779910
DOI: 10.1021/BI047709T
Last articles
Al in 7OL3Al in 7OP1
Al in 7OHN
Al in 7NVN
Al in 7NVM
Al in 7OH6
Al in 7NVL
Al in 7OH5
Al in 7OAR
Al in 7NVO