Zinc »
PDB 1y8q-1ykf »
1ycm »
Zinc in PDB 1ycm: Solution Structure of Matrix Metalloproteinase 12 (MMP12) in the Presence of N-Isobutyl-N-[4- Methoxyphenylsulfonyl]Glycyl Hydroxamic Acid (Nngh)
Enzymatic activity of Solution Structure of Matrix Metalloproteinase 12 (MMP12) in the Presence of N-Isobutyl-N-[4- Methoxyphenylsulfonyl]Glycyl Hydroxamic Acid (Nngh)
All present enzymatic activity of Solution Structure of Matrix Metalloproteinase 12 (MMP12) in the Presence of N-Isobutyl-N-[4- Methoxyphenylsulfonyl]Glycyl Hydroxamic Acid (Nngh):
3.4.24.65;
3.4.24.65;
Other elements in 1ycm:
The structure of Solution Structure of Matrix Metalloproteinase 12 (MMP12) in the Presence of N-Isobutyl-N-[4- Methoxyphenylsulfonyl]Glycyl Hydroxamic Acid (Nngh) also contains other interesting chemical elements:
| Calcium | (Ca) | 60 atoms |
Zinc Binding Sites:
The binding sites of Zinc atom in the Solution Structure of Matrix Metalloproteinase 12 (MMP12) in the Presence of N-Isobutyl-N-[4- Methoxyphenylsulfonyl]Glycyl Hydroxamic Acid (Nngh)
(pdb code 1ycm). This binding sites where shown within
5.0 Angstroms radius around Zinc atom.
In total 2 binding sites of Zinc where determined in the Solution Structure of Matrix Metalloproteinase 12 (MMP12) in the Presence of N-Isobutyl-N-[4- Methoxyphenylsulfonyl]Glycyl Hydroxamic Acid (Nngh), PDB code: 1ycm:
Jump to Zinc binding site number: 1; 2;
In total 2 binding sites of Zinc where determined in the Solution Structure of Matrix Metalloproteinase 12 (MMP12) in the Presence of N-Isobutyl-N-[4- Methoxyphenylsulfonyl]Glycyl Hydroxamic Acid (Nngh), PDB code: 1ycm:
Jump to Zinc binding site number: 1; 2;
Zinc binding site 1 out of 2 in 1ycm
Go back to
Zinc binding site 1 out
of 2 in the Solution Structure of Matrix Metalloproteinase 12 (MMP12) in the Presence of N-Isobutyl-N-[4- Methoxyphenylsulfonyl]Glycyl Hydroxamic Acid (Nngh)
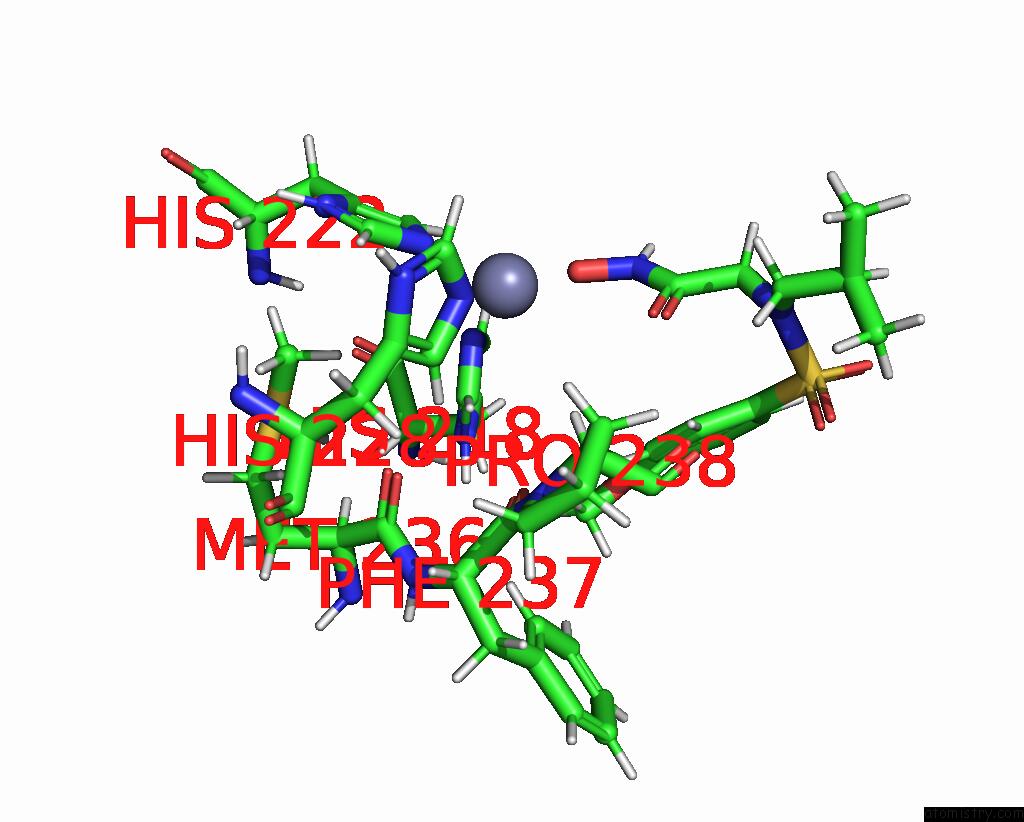
Mono view
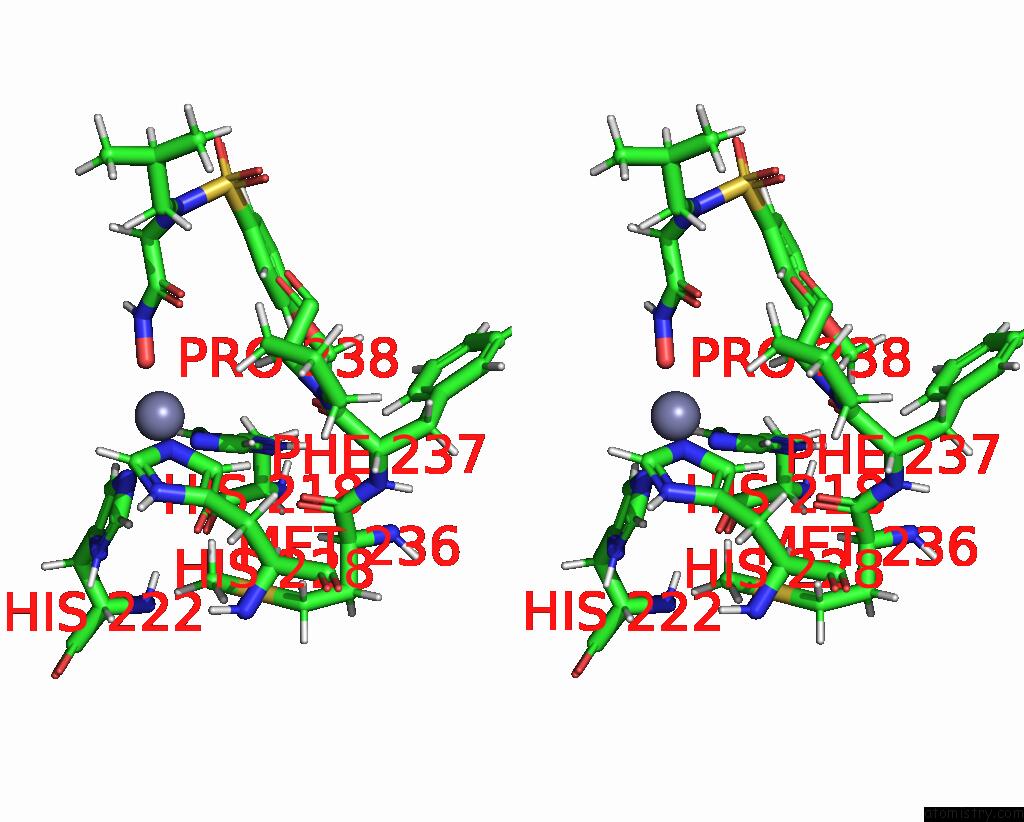
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 1 of Solution Structure of Matrix Metalloproteinase 12 (MMP12) in the Presence of N-Isobutyl-N-[4- Methoxyphenylsulfonyl]Glycyl Hydroxamic Acid (Nngh) within 5.0Å range:
|
Zinc binding site 2 out of 2 in 1ycm
Go back to
Zinc binding site 2 out
of 2 in the Solution Structure of Matrix Metalloproteinase 12 (MMP12) in the Presence of N-Isobutyl-N-[4- Methoxyphenylsulfonyl]Glycyl Hydroxamic Acid (Nngh)
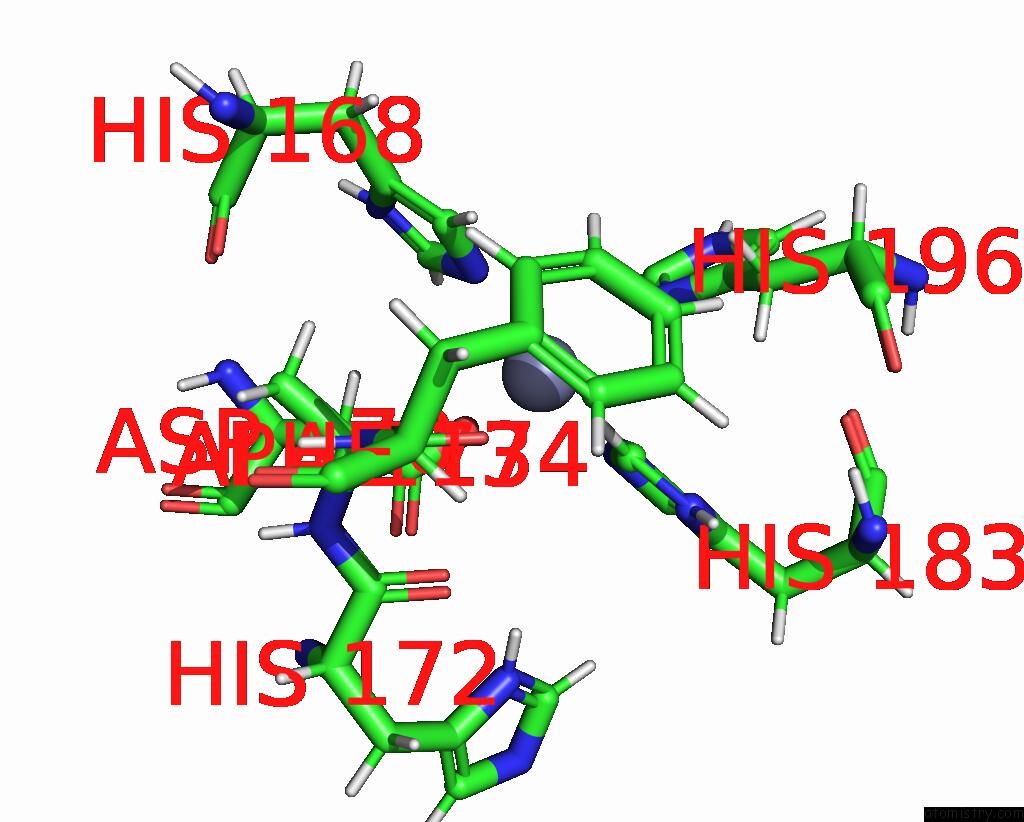
Mono view
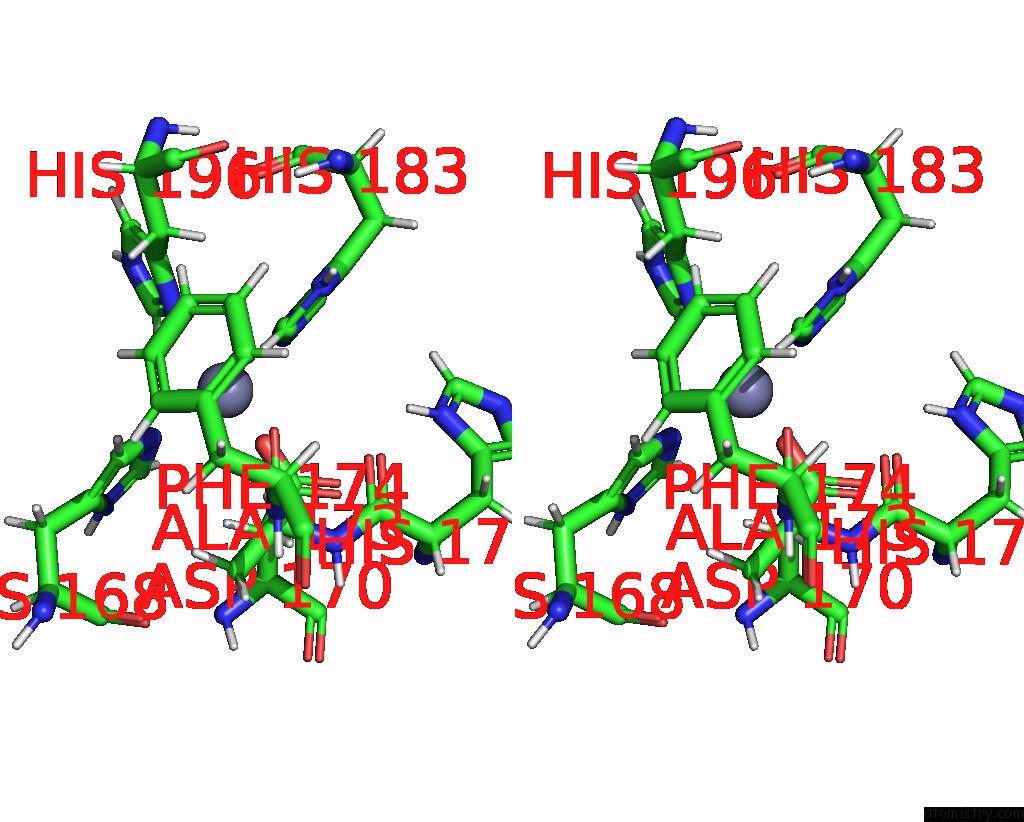
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 2 of Solution Structure of Matrix Metalloproteinase 12 (MMP12) in the Presence of N-Isobutyl-N-[4- Methoxyphenylsulfonyl]Glycyl Hydroxamic Acid (Nngh) within 5.0Å range:
|
Reference:
I.Bertini,
V.Calderone,
M.Cosenza,
M.Fragai,
Y.M.Lee,
C.Luchinat,
S.Mangani,
B.Terni,
P.Turano.
Conformational Variability of Matrix Metalloproteinases: Beyond A Single 3D Structure. Proc.Natl.Acad.Sci.Usa V. 102 5334 2005.
ISSN: ISSN 0027-8424
PubMed: 15809432
DOI: 10.1073/PNAS.0407106102
Page generated: Wed Oct 16 20:50:54 2024
ISSN: ISSN 0027-8424
PubMed: 15809432
DOI: 10.1073/PNAS.0407106102
Last articles
Zn in 9J0NZn in 9J0O
Zn in 9J0P
Zn in 9FJX
Zn in 9EKB
Zn in 9C0F
Zn in 9CAH
Zn in 9CH0
Zn in 9CH3
Zn in 9CH1