Zinc »
PDB 1tbt-1to4 »
1tno »
Zinc in PDB 1tno: Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B
Enzymatic activity of Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B
All present enzymatic activity of Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B:
2.5.1.59;
2.5.1.59;
Protein crystallography data
The structure of Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B, PDB code: 1tno
was solved by
T.S.Reid,
K.L.Terry,
P.J.Casey,
L.S.Beese,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 29.98 / 2.70 |
| Space group | C 1 2 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 272.531, 268.484, 185.940, 90.00, 131.48, 90.00 |
| R / Rfree (%) | 19.4 / 21.1 |
Other elements in 1tno:
The structure of Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B also contains other interesting chemical elements:
| Chlorine | (Cl) | 4 atoms |
Zinc Binding Sites:
The binding sites of Zinc atom in the Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B
(pdb code 1tno). This binding sites where shown within
5.0 Angstroms radius around Zinc atom.
In total 6 binding sites of Zinc where determined in the Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B, PDB code: 1tno:
Jump to Zinc binding site number: 1; 2; 3; 4; 5; 6;
In total 6 binding sites of Zinc where determined in the Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B, PDB code: 1tno:
Jump to Zinc binding site number: 1; 2; 3; 4; 5; 6;
Zinc binding site 1 out of 6 in 1tno
Go back to
Zinc binding site 1 out
of 6 in the Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B
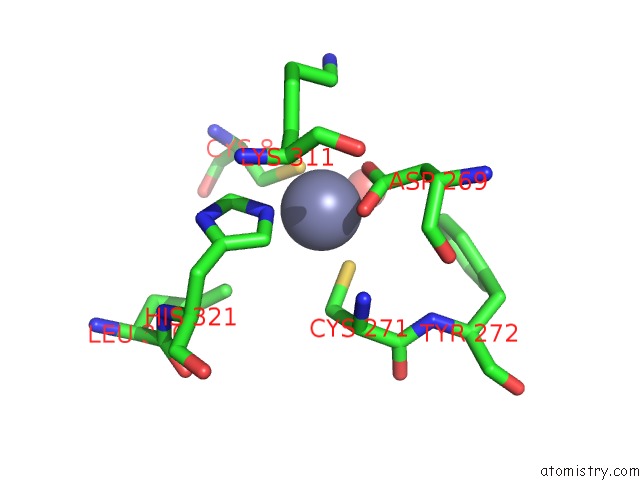
Mono view

Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 1 of Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B within 5.0Å range:
|
Zinc binding site 2 out of 6 in 1tno
Go back to
Zinc binding site 2 out
of 6 in the Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B
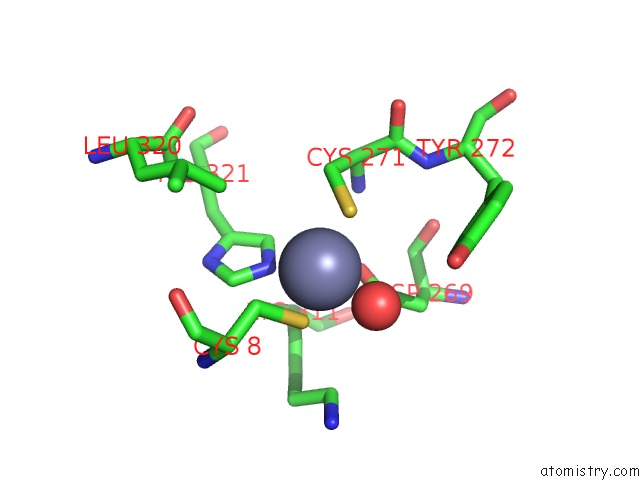
Mono view
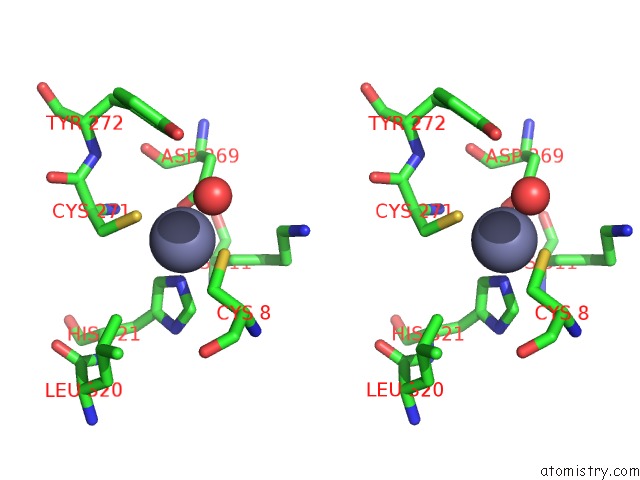
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 2 of Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B within 5.0Å range:
|
Zinc binding site 3 out of 6 in 1tno
Go back to
Zinc binding site 3 out
of 6 in the Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B
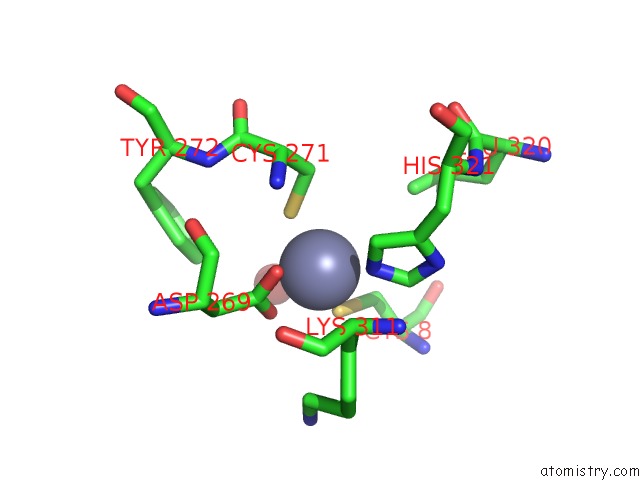
Mono view
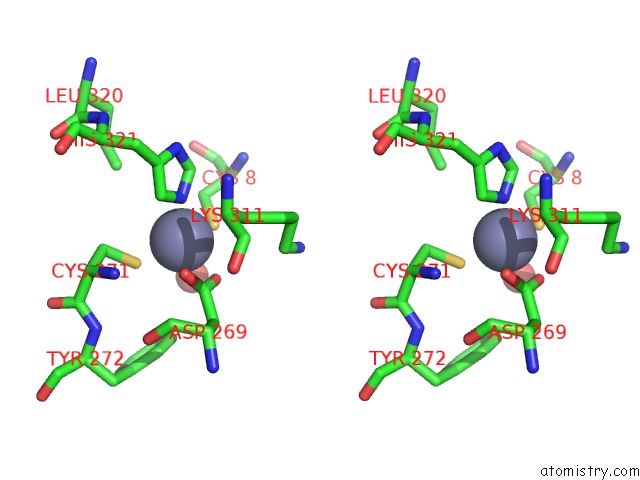
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 3 of Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B within 5.0Å range:
|
Zinc binding site 4 out of 6 in 1tno
Go back to
Zinc binding site 4 out
of 6 in the Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B

Mono view

Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 4 of Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B within 5.0Å range:
|
Zinc binding site 5 out of 6 in 1tno
Go back to
Zinc binding site 5 out
of 6 in the Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B

Mono view
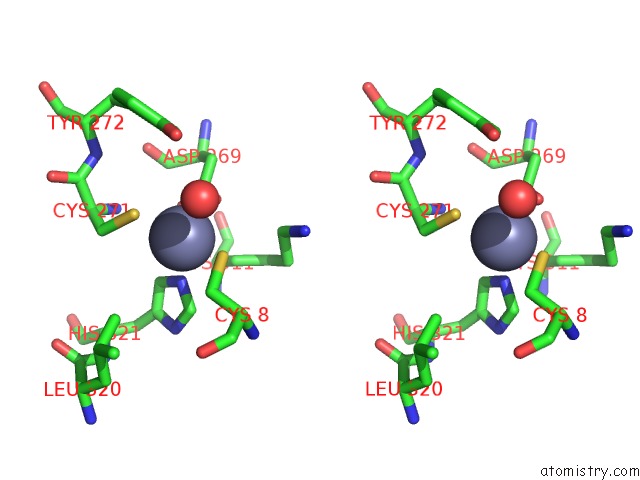
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 5 of Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B within 5.0Å range:
|
Zinc binding site 6 out of 6 in 1tno
Go back to
Zinc binding site 6 out
of 6 in the Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B
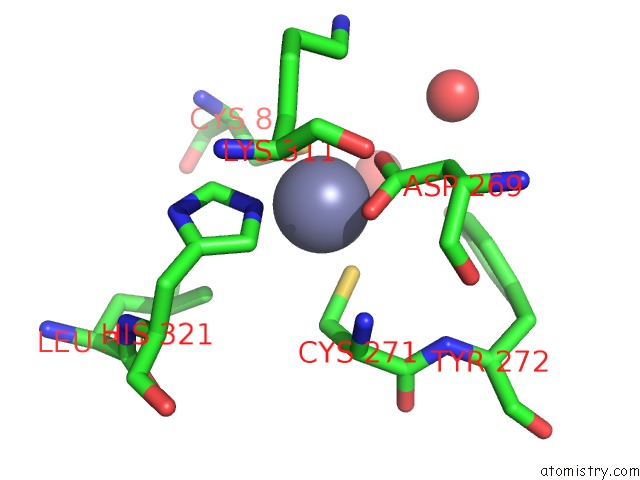
Mono view
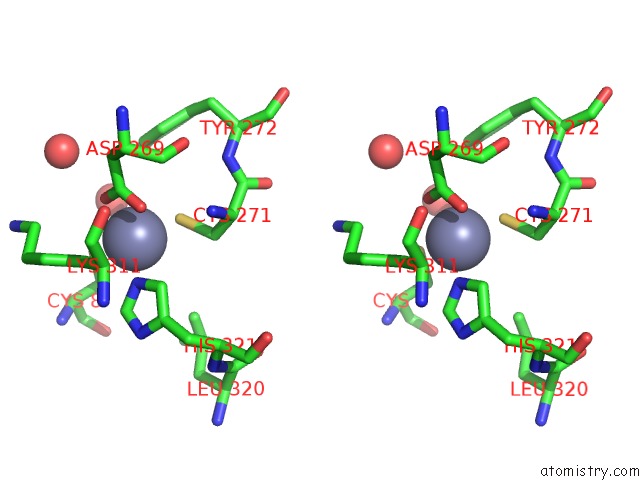
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 6 of Rat Protein Geranylgeranyltransferase Type-I Complexed with A Ggpp Analog and A Kkksktkcvim Peptide Derived From K- RAS4B within 5.0Å range:
|
Reference:
T.S.Reid,
K.L.Terry,
P.J.Casey,
L.S.Beese.
Crystallographic Analysis of Caax Prenyltransferases Complexed with Substrates Defines Rules of Protein Substrate Selectivity. J.Mol.Biol. V. 343 417 2004.
ISSN: ISSN 0022-2836
PubMed: 15451670
DOI: 10.1016/J.JMB.2004.08.056
Page generated: Wed Oct 16 19:12:12 2024
ISSN: ISSN 0022-2836
PubMed: 15451670
DOI: 10.1016/J.JMB.2004.08.056
Last articles
Zn in 9J0NZn in 9J0O
Zn in 9J0P
Zn in 9FJX
Zn in 9EKB
Zn in 9C0F
Zn in 9CAH
Zn in 9CH0
Zn in 9CH3
Zn in 9CH1