Zinc »
PDB 7aof-7av1 »
7apm »
Zinc in PDB 7apm: Trna-Guanine Transglycosylase H319C Mutant Spin-Labeled with Mtsl
Enzymatic activity of Trna-Guanine Transglycosylase H319C Mutant Spin-Labeled with Mtsl
All present enzymatic activity of Trna-Guanine Transglycosylase H319C Mutant Spin-Labeled with Mtsl:
2.4.2.29;
2.4.2.29;
Protein crystallography data
The structure of Trna-Guanine Transglycosylase H319C Mutant Spin-Labeled with Mtsl, PDB code: 7apm
was solved by
D.Nguyen,
A.Heine,
G.Klebe,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 45.68 / 1.66 |
| Space group | C 1 2 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 91.911, 65.064, 70.813, 90.00, 96.29, 90.00 |
| R / Rfree (%) | 13.9 / 17.6 |
Other elements in 7apm:
The structure of Trna-Guanine Transglycosylase H319C Mutant Spin-Labeled with Mtsl also contains other interesting chemical elements:
| Chlorine | (Cl) | 1 atom |
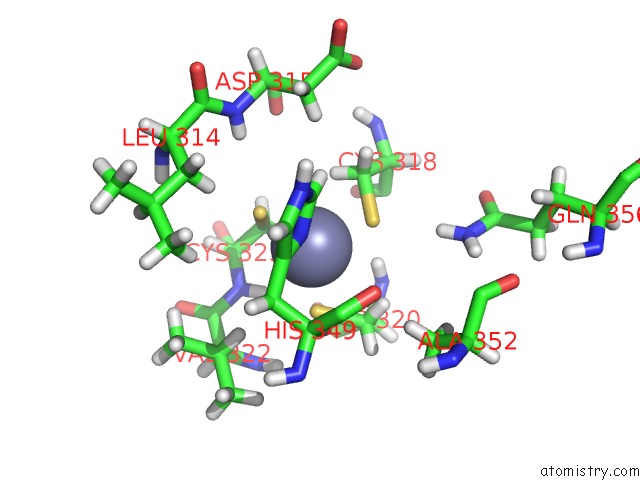
Zinc Binding Sites:
The binding sites of Zinc atom in the Trna-Guanine Transglycosylase H319C Mutant Spin-Labeled with Mtsl
(pdb code 7apm). This binding sites where shown within
5.0 Angstroms radius around Zinc atom.
In total only one binding site of Zinc was determined in the Trna-Guanine Transglycosylase H319C Mutant Spin-Labeled with Mtsl, PDB code: 7apm:
In total only one binding site of Zinc was determined in the Trna-Guanine Transglycosylase H319C Mutant Spin-Labeled with Mtsl, PDB code: 7apm:
Zinc binding site 1 out of 1 in 7apm
Go back to
Zinc binding site 1 out
of 1 in the Trna-Guanine Transglycosylase H319C Mutant Spin-Labeled with Mtsl
Mono view
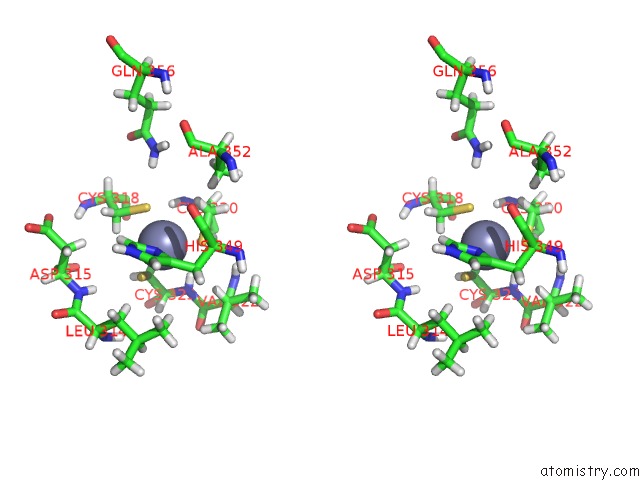
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 1 of Trna-Guanine Transglycosylase H319C Mutant Spin-Labeled with Mtsl within 5.0Å range:
|
Reference:
D.Abdullin,
D.Nguyen,
C.A.Heubach,
A.Nguyen,
T.Pfaffeneder,
A.Heine,
K.Reuter,
F.Diederich,
O.Schiemann,
G.Klebe.
Unraveling A Ligand-Induced Twist of A Homodimeric Enzyme By Pulsed Electron-Electron Double Resonance To Be Published.
Page generated: Tue Oct 29 17:05:28 2024
Last articles
As in 3FKGAs in 3FM4
As in 3FMU
As in 3ET6
As in 3ENZ
As in 3FJU
As in 3FCU
As in 3F7D
As in 3ERP
As in 3E7S