Zinc »
PDB 6ngn-6nla »
6nj2 »
Zinc in PDB 6nj2: Thermostable Carbonic Anhydrase II Variant with Tetrazine 2.0 at Site 186
Enzymatic activity of Thermostable Carbonic Anhydrase II Variant with Tetrazine 2.0 at Site 186
All present enzymatic activity of Thermostable Carbonic Anhydrase II Variant with Tetrazine 2.0 at Site 186:
4.2.1.1;
4.2.1.1;
Protein crystallography data
The structure of Thermostable Carbonic Anhydrase II Variant with Tetrazine 2.0 at Site 186, PDB code: 6nj2
was solved by
K.M.Kean,
P.A.Karplus,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 37.25 / 1.50 |
| Space group | P 21 21 21 |
| Cell size a, b, c (Å), α, β, γ (°) | 61.440, 93.680, 101.160, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 17.2 / 19.6 |
Zinc Binding Sites:
The binding sites of Zinc atom in the Thermostable Carbonic Anhydrase II Variant with Tetrazine 2.0 at Site 186
(pdb code 6nj2). This binding sites where shown within
5.0 Angstroms radius around Zinc atom.
In total 2 binding sites of Zinc where determined in the Thermostable Carbonic Anhydrase II Variant with Tetrazine 2.0 at Site 186, PDB code: 6nj2:
Jump to Zinc binding site number: 1; 2;
In total 2 binding sites of Zinc where determined in the Thermostable Carbonic Anhydrase II Variant with Tetrazine 2.0 at Site 186, PDB code: 6nj2:
Jump to Zinc binding site number: 1; 2;
Zinc binding site 1 out of 2 in 6nj2
Go back to
Zinc binding site 1 out
of 2 in the Thermostable Carbonic Anhydrase II Variant with Tetrazine 2.0 at Site 186
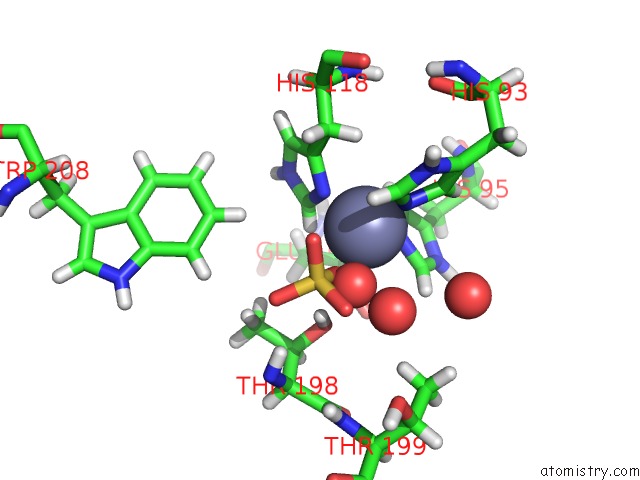
Mono view
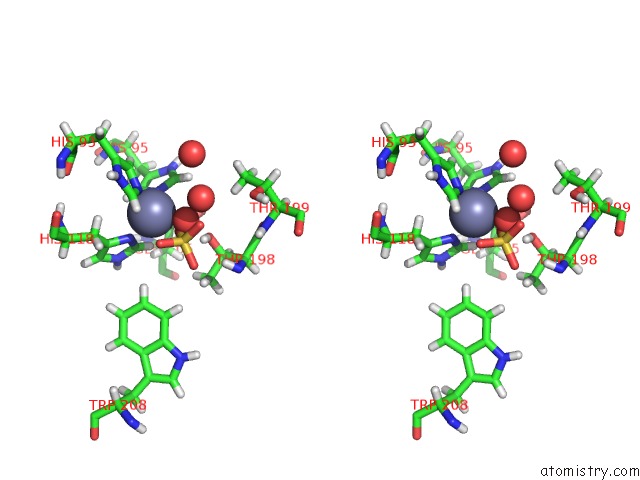
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 1 of Thermostable Carbonic Anhydrase II Variant with Tetrazine 2.0 at Site 186 within 5.0Å range:
|
Zinc binding site 2 out of 2 in 6nj2
Go back to
Zinc binding site 2 out
of 2 in the Thermostable Carbonic Anhydrase II Variant with Tetrazine 2.0 at Site 186
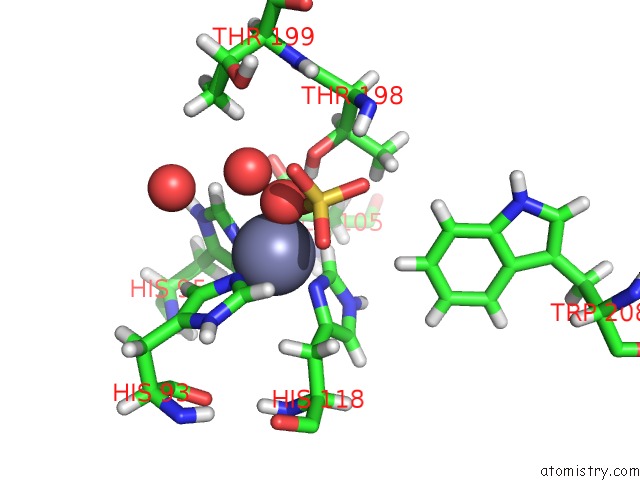
Mono view
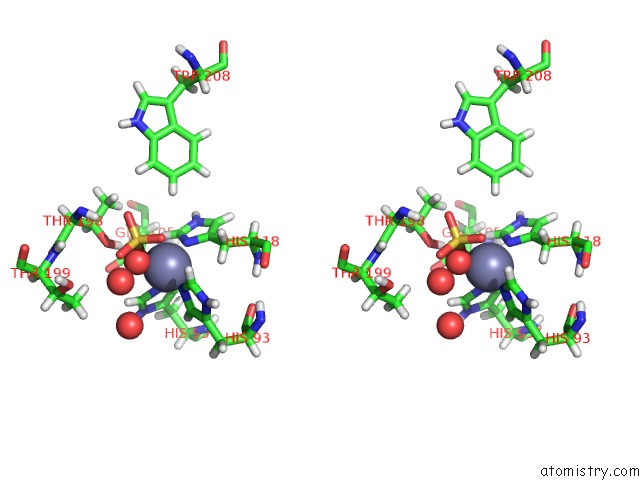
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 2 of Thermostable Carbonic Anhydrase II Variant with Tetrazine 2.0 at Site 186 within 5.0Å range:
|
Reference:
R.M.Bednar,
T.W.Golbek,
K.M.Kean,
W.J.Brown,
S.Jana,
J.E.Baio,
P.A.Karplus,
R.A.Mehl.
Immobilization of Proteins with Controlled Load and Orientation. Acs Appl Mater Interfaces V. 11 36391 2019.
ISSN: ISSN 1944-8252
PubMed: 31525993
DOI: 10.1021/ACSAMI.9B12746
Page generated: Thu Aug 21 17:49:17 2025
ISSN: ISSN 1944-8252
PubMed: 31525993
DOI: 10.1021/ACSAMI.9B12746
Last articles
Zn in 6X68Zn in 6X67
Zn in 6X5A
Zn in 6X5X
Zn in 6WVV
Zn in 6X50
Zn in 6X59
Zn in 6X4Y
Zn in 6X4W
Zn in 6X46