Zinc »
PDB 1r2y-1raf »
1r43 »
Zinc in PDB 1r43: Crystal Structure of Beta-Alanine Synthase From Saccharomyces Kluyveri (Selenomethionine Substituted Protein)
Enzymatic activity of Crystal Structure of Beta-Alanine Synthase From Saccharomyces Kluyveri (Selenomethionine Substituted Protein)
All present enzymatic activity of Crystal Structure of Beta-Alanine Synthase From Saccharomyces Kluyveri (Selenomethionine Substituted Protein):
3.5.1.6;
3.5.1.6;
Protein crystallography data
The structure of Crystal Structure of Beta-Alanine Synthase From Saccharomyces Kluyveri (Selenomethionine Substituted Protein), PDB code: 1r43
was solved by
S.Lundgren,
Z.Gojkovic,
J.Piskur,
D.Dobritzsch,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 29.75 / 2.80 |
| Space group | P 1 21 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 60.430, 77.630, 110.570, 90.00, 95.63, 90.00 |
| R / Rfree (%) | 21.1 / 27.4 |
Zinc Binding Sites:
The binding sites of Zinc atom in the Crystal Structure of Beta-Alanine Synthase From Saccharomyces Kluyveri (Selenomethionine Substituted Protein)
(pdb code 1r43). This binding sites where shown within
5.0 Angstroms radius around Zinc atom.
In total 4 binding sites of Zinc where determined in the Crystal Structure of Beta-Alanine Synthase From Saccharomyces Kluyveri (Selenomethionine Substituted Protein), PDB code: 1r43:
Jump to Zinc binding site number: 1; 2; 3; 4;
In total 4 binding sites of Zinc where determined in the Crystal Structure of Beta-Alanine Synthase From Saccharomyces Kluyveri (Selenomethionine Substituted Protein), PDB code: 1r43:
Jump to Zinc binding site number: 1; 2; 3; 4;
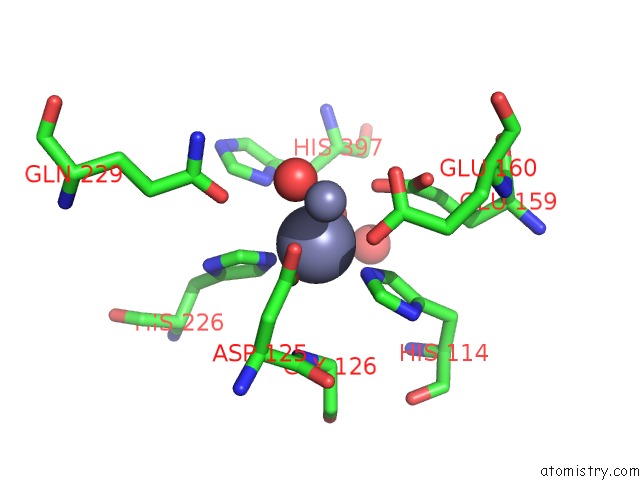
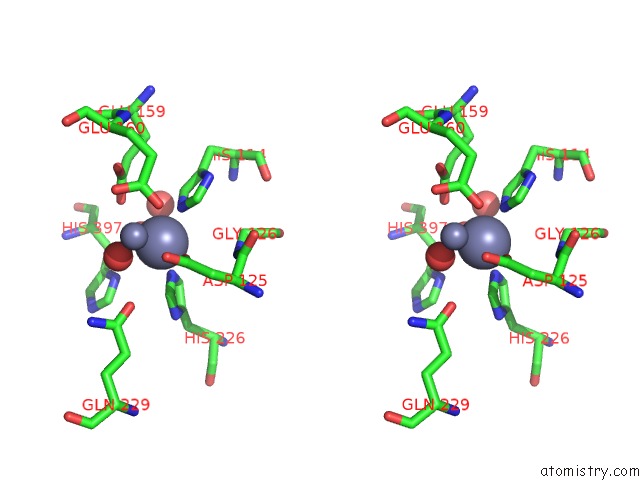
Zinc binding site 1 out of 4 in 1r43
Go back to
Zinc binding site 1 out
of 4 in the Crystal Structure of Beta-Alanine Synthase From Saccharomyces Kluyveri (Selenomethionine Substituted Protein)
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 1 of Crystal Structure of Beta-Alanine Synthase From Saccharomyces Kluyveri (Selenomethionine Substituted Protein) within 5.0Å range:
|
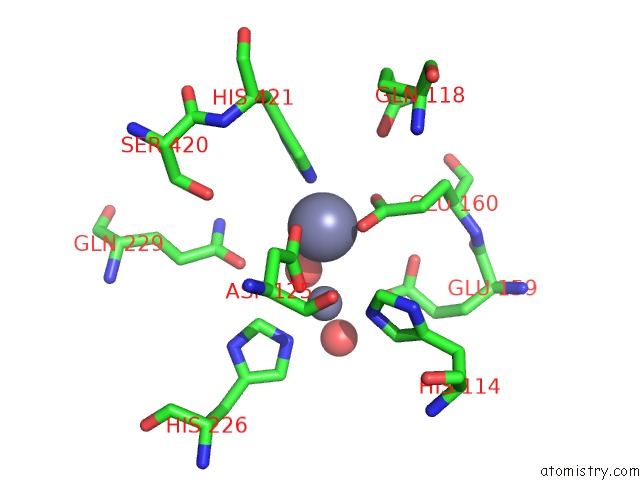
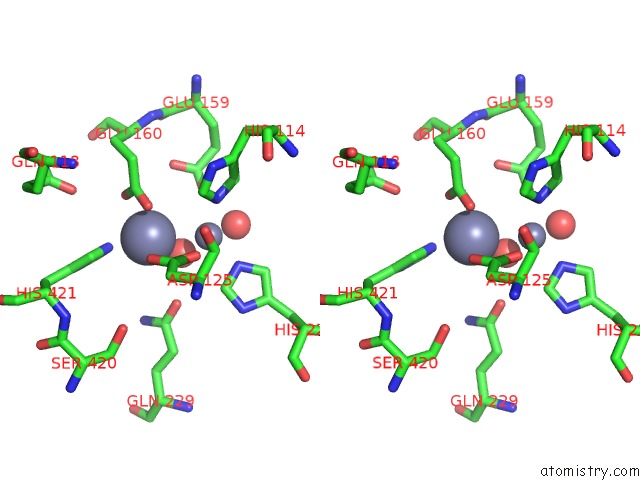
Zinc binding site 2 out of 4 in 1r43
Go back to
Zinc binding site 2 out
of 4 in the Crystal Structure of Beta-Alanine Synthase From Saccharomyces Kluyveri (Selenomethionine Substituted Protein)
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 2 of Crystal Structure of Beta-Alanine Synthase From Saccharomyces Kluyveri (Selenomethionine Substituted Protein) within 5.0Å range:
|
Zinc binding site 3 out of 4 in 1r43
Go back to
Zinc binding site 3 out
of 4 in the Crystal Structure of Beta-Alanine Synthase From Saccharomyces Kluyveri (Selenomethionine Substituted Protein)
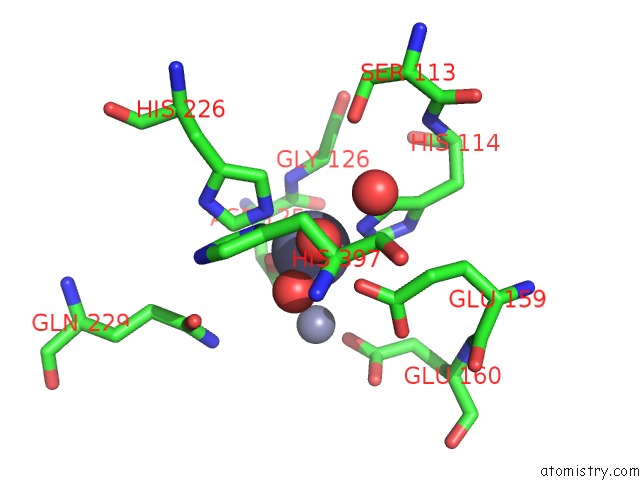
Mono view
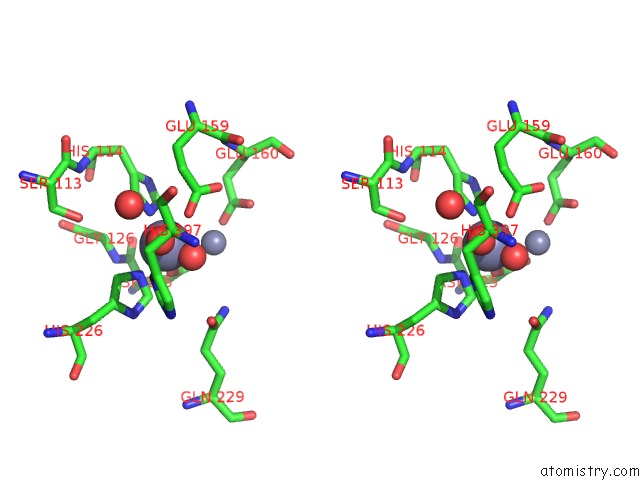
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 3 of Crystal Structure of Beta-Alanine Synthase From Saccharomyces Kluyveri (Selenomethionine Substituted Protein) within 5.0Å range:
|
Zinc binding site 4 out of 4 in 1r43
Go back to
Zinc binding site 4 out
of 4 in the Crystal Structure of Beta-Alanine Synthase From Saccharomyces Kluyveri (Selenomethionine Substituted Protein)
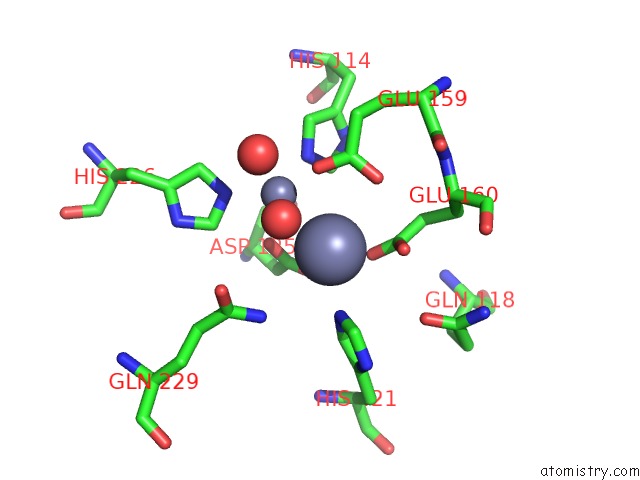
Mono view
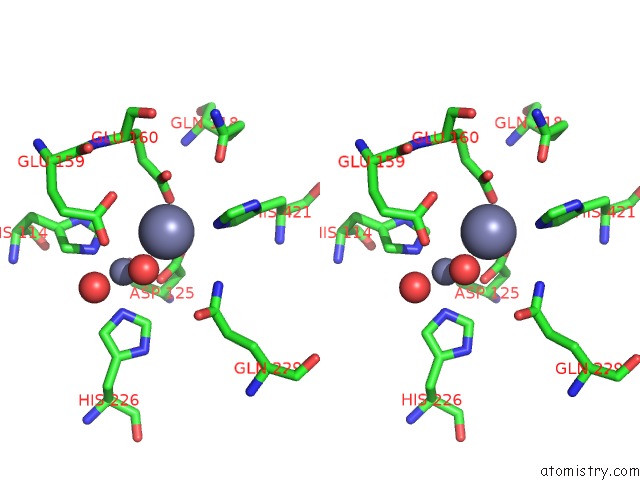
Stereo pair view

Mono view

Stereo pair view
A full contact list of Zinc with other atoms in the Zn binding
site number 4 of Crystal Structure of Beta-Alanine Synthase From Saccharomyces Kluyveri (Selenomethionine Substituted Protein) within 5.0Å range:
|
Reference:
S.Lundgren,
Z.Gojkovic,
J.Piskur,
D.Dobritzsch.
Yeast Beta-Alanine Synthase Shares A Structural Scaffold and Origin with Dizinc-Dependent Exopeptidases J.Biol.Chem. V. 278 51851.
ISSN: ISSN 0021-9258
PubMed: 14534321
DOI: 10.1074/JBC.M308674200
Page generated: Wed Oct 16 18:21:10 2024
ISSN: ISSN 0021-9258
PubMed: 14534321
DOI: 10.1074/JBC.M308674200
Last articles
As in 3N68As in 3N67
As in 3N6A
As in 3N69
As in 3N5T
As in 3N5S
As in 3N5R
As in 3M1R
As in 3N5Q
As in 3N5P